Difference between revisions of "2017 DOCK tutorial 2 with PDB 3GPL NEW"
Stonybrook (talk | contribs) (→III. Generating Receptor Surface and Spheres) |
Stonybrook (talk | contribs) (→3PGL) |
||
| (19 intermediate revisions by the same user not shown) | |||
| Line 15: | Line 15: | ||
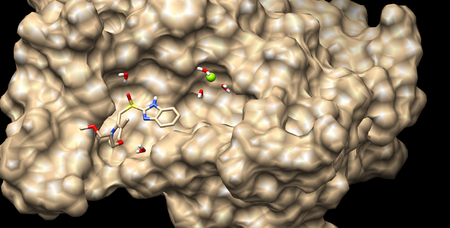
| − | In this tutorial we will use PDB code 3PGL, the deposited crystal structure of | + | In this tutorial we will use PDB code 3PGL, the deposited crystal structure of human small C-terminal domain phosphatase 1 in complex with rabeprazole. |
| − | [[Image:2017_3pgl_receptor_surface.png|thumb|center|450px| | + | [[Image:2017_3pgl_receptor_surface.png|thumb|center|450px| 3pgl in complex with rabeprazole. Waters and Magnesium ion in binding pocket are shown]] |
===Organizing Directories=== | ===Organizing Directories=== | ||
| Line 39: | Line 39: | ||
==II. Preparing the Receptor and Ligand== | ==II. Preparing the Receptor and Ligand== | ||
| + | |||
| + | One issue that you will encounter when running Dock using the standard procedure on this system is that the ligand will move greatly. This is because normally all waters in the crystal structure are deleted, but they seem to be important for the positioning of the ligand. That causes the procedure in this tutorial to be slightly different from previous tutorials. | ||
Download the PDB file (3PGL) and move file 3PGL.pdb to 00.files | Download the PDB file (3PGL) and move file 3PGL.pdb to 00.files | ||
| Line 47: | Line 49: | ||
raw_3pgl.pdb was opened with VI terminal editor | raw_3pgl.pdb was opened with VI terminal editor | ||
| − | + | Delete the header information, connect records, and waters except for waters 2, 21, 32, 39, and 258 (these numbers are listed in the 6th column of the dpb file). Change HETATM to ATOM, and delete all rows that have B in the fifth column | |
| + | Change RZX A to LIG B to make the ligand a different chain | ||
| − | + | Now open the system in chimera and use | |
| + | Tools-->Structure Editing-->Add Charge | ||
| + | Select AMBER ff99SB for Standard Residues, and add a charge of +2 to Mg and 0 to everything else | ||
'''Prepare ligand and receptor files for dock''' | '''Prepare ligand and receptor files for dock''' | ||
| − | + | All waters were deleted from the file except for the ones believed to interact with the ligand and the Mg atom. These are believed to be important for the proper binding of the ligand. To properly orient the waters, run the system through tleap, keeping everything fixed except the 5 water molecules (this process can be seen in the amber tutorials on this wiki). This new system will yield better docking results, and should be saved as 3pgl.dockprep.mol2. | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | Open 3pgl.dockprep.mol2 in Chimera. Select the ligand and click Actions-->Atoms/Bonds-->delete. Save this file as 3pgl.rec.mol2. | |
| − | + | With just the receptor open in Chimera, go to Select-->Chemistry-->element-->H and click Actions-->Atoms/Bonds-->delete. Save this file as 3pgl.rec.noH.mol2 | |
| − | + | Open 3pgl.dockprep.mol2 in Chimera and select the receptor. Click Actions-->Atoms/Bonds-->delete to get rid of everything except the ligand. Save this as 3pgl.lig.mol2. | |
==III. Generating Receptor Surface and Spheres== | ==III. Generating Receptor Surface and Spheres== | ||
| Line 121: | Line 116: | ||
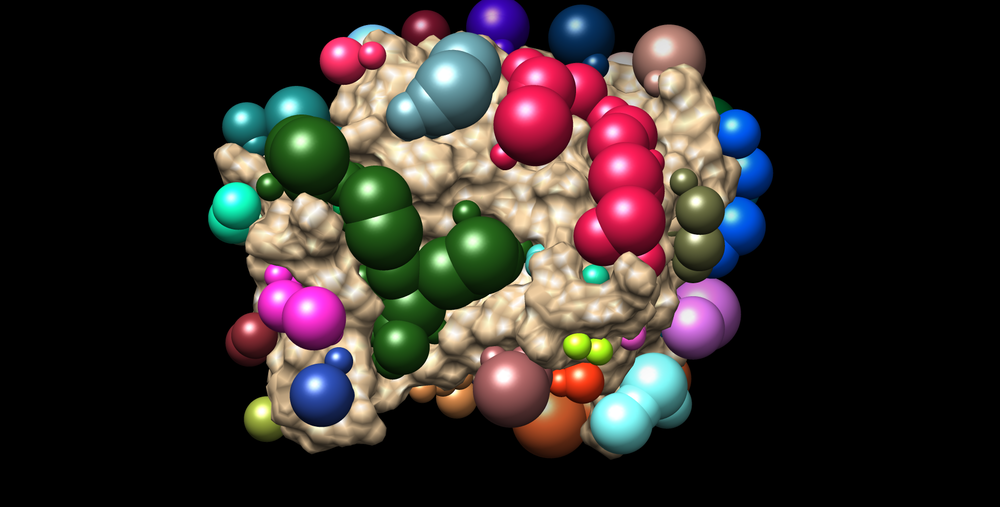
The image that appears should resemble this: | The image that appears should resemble this: | ||
| − | [[Image:3pgl_all_spheres.png|thumb|center|1000px| | + | [[Image:3pgl_all_spheres.png|thumb|center|1000px|3PGL receptor with all spheres.]] |
| Line 138: | Line 133: | ||
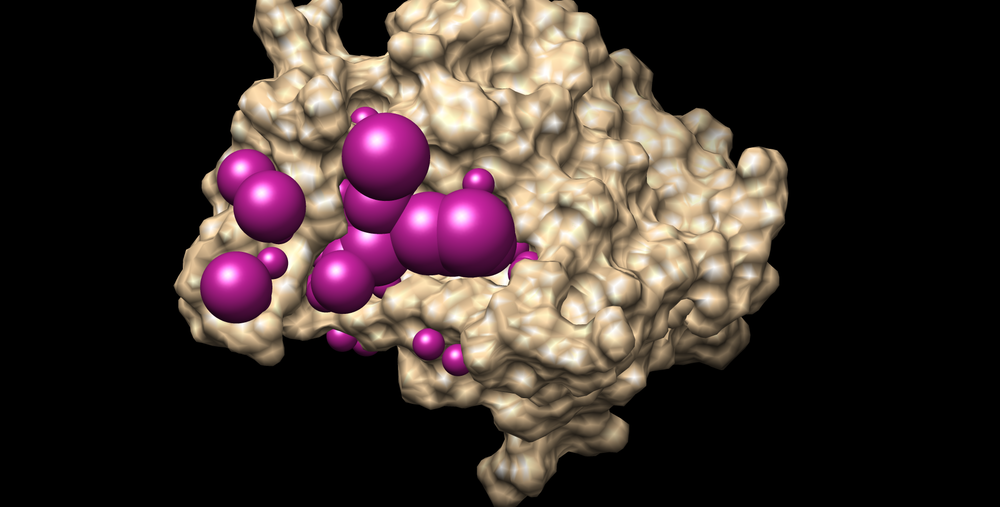
The selected spheres with the receptor surface should look similar to that as seen below: | The selected spheres with the receptor surface should look similar to that as seen below: | ||
| − | [[Image: | + | [[Image:3pgl_selected_spheres.png|thumb|center|1000px|3PGL receptor surface with spheres within 8.0 Angstroms of known ligand]] |
==IV. Generating Box and Grid== | ==IV. Generating Box and Grid== | ||
| Line 168: | Line 163: | ||
Compute the energy grid | Compute the energy grid | ||
| − | create grid.in | + | create grid.in and grid out files using |
touch grid.in | touch grid.in | ||
| + | touch grid.out | ||
| + | |||
| + | |||
| + | |||
| + | Compute the grid, and save the output in the file grid.out | ||
| + | grid -i grid.in > grid.out | ||
When prpmped, answer the questions as follows: | When prpmped, answer the questions as follows: | ||
| Line 267: | Line 268: | ||
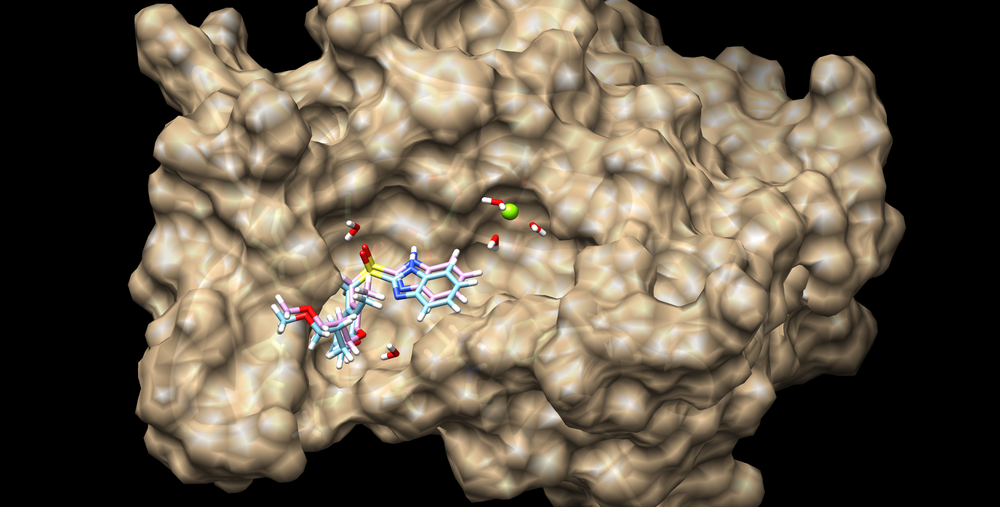
This structure can be visualized using chimera and loading the surface into chimera along with the minimized and unminimized ligand structures: | This structure can be visualized using chimera and loading the surface into chimera along with the minimized and unminimized ligand structures: | ||
| − | [[Image: | + | [[Image:01_minimized_lig.png|thumb|center|1000px|3pgl with ligand before and after minimization]] |
===Footprint Calculation=== | ===Footprint Calculation=== | ||
| Line 345: | Line 346: | ||
The output will look something like this: | The output will look something like this: | ||
| − | [[Image: | + | [[Image:Fps_lig_minlig.png|thumb|center|1000px|Footprint comparison between unminimized and minimized ligand structure]] |
===Rigid Docking=== | ===Rigid Docking=== | ||
| Line 425: | Line 426: | ||
The best scoring result should look like | The best scoring result should look like | ||
| − | + | [[Image:02_rigid_bestscore.png|thumb|center|800px|Rigid docking results]] | |
===Flexible Docking=== | ===Flexible Docking=== | ||
| Line 515: | Line 516: | ||
These can be viewed in the same way as before and the best scoring result should look like | These can be viewed in the same way as before and the best scoring result should look like | ||
| − | + | [[Image:03_flex_bestscore.png|thumb|center|800px|Flexible docking results]] | |
===Fixed Anchor Docking=== | ===Fixed Anchor Docking=== | ||
| Line 603: | Line 604: | ||
cluster_rmsd_threshold 2.0 | cluster_rmsd_threshold 2.0 | ||
rank_ligands no | rank_ligands no | ||
| + | |||
| + | The top scoring molecule is pictured below | ||
| + | [[Image:04_fad_bestscore.png|thumb|center|800px|Fixed Anchor Docking results]] | ||
==VI. Virtual Screening== | ==VI. Virtual Screening== | ||
| + | At this point we will begin docking potential inhibitors into the active site of 4qmz. The compound library we will use contains 25,000 molecules and was previously prepared for another docking experiment. The compound library contains molecules with rotatable bonds number between 7 and 15. The library used is titled small_ligand_library.mol2, this library can be found at /gpfs/projects/AMS536/virtual_screen_library on seawulf. | ||
| + | |||
| + | We began by copying all of our local directories outlined in the first section to a project directory on seawulf to allow use to expedite the docking process over the 25,000 compounds in our test library. | ||
| + | |||
| + | We will create a virtual-screen.in file that looks like the following: | ||
| + | |||
| + | conformer_search_type flex | ||
| + | user_specified_anchor no | ||
| + | limit_max_anchors no | ||
| + | min_anchor_size 5 | ||
| + | pruning_use_clustering yes | ||
| + | pruning_max_orients 1000 | ||
| + | pruning_clustering_cutoff 100 | ||
| + | pruning_conformer_score_cutoff 100.0 | ||
| + | pruning_conformer_score_scaling_factor 1.0 | ||
| + | use_clash_overlap no | ||
| + | write_growth_tree no | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file small_ligand_library.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd yes | ||
| + | use_rmsd_reference_mol yes | ||
| + | rmsd_reference_filename ../01.dockprep/4qmz.lig.mol2 | ||
| + | use_database_filter no | ||
| + | orient_ligand yes | ||
| + | automated_matching yes | ||
| + | receptor_site_file ../02.surface-spheres/selected_spheres.sph | ||
| + | max_orientations 1000 | ||
| + | critical_points no | ||
| + | chemical_matching no | ||
| + | use_ligand_spheres no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix ../03.box-grid/grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | minimize_anchor yes | ||
| + | minimize_flexible_growth yes | ||
| + | use_advanced_simplex_parameters no | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_anchor_max_iterations 500 | ||
| + | simplex_grow_max_iterations 500 | ||
| + | simplex_grow_tors_premin_iterations 0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix 4qmz.virtualscreen | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | write_conformations no | ||
| + | cluster_conformations yes | ||
| + | cluster_rmsd_threshold 2.0 | ||
| + | rank_ligands no | ||
| + | |||
| + | This script is submitted to the seawulf cluster using a script called screen.sh which contains the following: | ||
| + | |||
| + | #! /bin/tcsh | ||
| + | #PBS -l nodes=4:ppn=28 | ||
| + | #PBS -l walltime=24:00:00 | ||
| + | #PBS -o jobstatus.out | ||
| + | #PBS -e joberror.err | ||
| + | #PBS -N large_vs | ||
| + | #PBS -V | ||
| + | #PBS -q long | ||
| + | cd $PBS_O_WORKDIR | ||
| + | mpirun -np 112 dock6.mpi -i virtual-screen.in -o virtualscreen1.out | ||
| + | |||
| + | |||
| + | This screen.sh input specifies that we will use 112 processors to dock our virtual library. We have a wall clock for the job of 24 hours and we will output jobstatus.out, joberror.err and virtualscreen1.out. In addition to these output files there is an output for the work performed by each processor individually. | ||
| + | |||
| + | ===Cartesian Minimization of the docked molecules=== | ||
| + | |||
| + | Now we will do cartesian minimization of the docked molecules. Cartesian here is referring to the receptor being explicitly present during the minimization. | ||
| + | |||
| + | Write the minimization input script minimize-vs.in: | ||
| + | |||
| + | conformer_search_type rigid | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file 4qmz.virtualscreen_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd no | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary no | ||
| + | grid_score_secondary no | ||
| + | multigrid_score_primary no | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_primary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_primary yes | ||
| + | continuous_score_secondary no | ||
| + | cont_score_rec_filename ../01.dockprep/4qmz.rec.mol2 | ||
| + | cont_score_att_exp 6 | ||
| + | cont_score_rep_exp 12 | ||
| + | cont_score_rep_rad_scale 1 | ||
| + | cont_score_use_dist_dep_dielectric yes | ||
| + | cont_score_dielectric 4.0 | ||
| + | cont_score_vdw_scale 1 | ||
| + | cont_score_es_scale 1 | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | simplex_max_iterations 1000 | ||
| + | simplex_tors_premin_iterations 0 | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix 4qmz.virtualscreen1.minimized | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | |||
| + | Submit the minimization job using with the script "job-min.sh" following lines: | ||
| + | |||
| + | #!/bin/tcsh | ||
| + | #PBS -l nodes=16:ppn=28 | ||
| + | #PBS -l walltime=12:00:00 | ||
| + | #PBS -o jobstatusmin.out | ||
| + | #PBS -e jobstatusmin.err | ||
| + | #PBS -N chunk0min | ||
| + | #PBS -V | ||
| + | #PBS -q medium | ||
| + | module load intel/mpi/64/2017/0.098 | ||
| + | cd $PBS_O_WORKDIR | ||
| + | mpirun -np 448 /gpfs/projects/AMS536/zzz.programs/dock6/bin/dock6.mpi -i minimize-vs.in -o minimize-vs.out | ||
| + | |||
| + | |||
| + | At this point it would be good to rank our docked ligands by score and extract the 100 best scoring (100 lowest scoring, most negative) ligands to investigate further. | ||
==VIII. Frequently Encountered Problems== | ==VIII. Frequently Encountered Problems== | ||
Latest revision as of 17:59, 8 March 2017
For additional Rizzo Lab tutorials see DOCK Tutorials. Use this link Wiki Formatting as a reference for editing the wiki. This tutorial was developed collaboratively by a subsection of the AMS 536 class of 2017, using DOCK v6.8.
Contents
I. Introduction
DOCK
DOCK is a molecular docking program used in drug discovery. It was developed by Irwin D. Kuntz, Jr. and colleagues at UCSF (see UCSF DOCK). This program, given a protein binding site and a small molecule, tries to predict the correct binding mode of the small molecule in the binding site, and the associated binding energy. Small molecules with highly favorable binding energies could be new drug leads. This makes DOCK a valuable drug discovery tool. DOCK is typically used to screen massive libraries of millions of compounds against a protein to isolate potential drug leads. These leads are then further studied, and could eventually result in a new, marketable drug. DOCK works well as a screening procedure for generating leads, but is not currently as useful for optimization of those leads.
DOCK 6 uses an incremental construction algorithm called anchor and grow. It is described by a three-step process:
- Rigid portion of ligand (anchor) is docked by geometric methods.
- Non-rigid segments added in layers; energy minimized.
- The resulting configurations are 'pruned' and energy re-minimized, yielding the docked configurations.
3PGL
In this tutorial we will use PDB code 3PGL, the deposited crystal structure of human small C-terminal domain phosphatase 1 in complex with rabeprazole.
Organizing Directories
While performing docking, it is convenient to adopt a standard directory structure / naming scheme, so that files are easy to find / identify.For this tutorial, we will use something similar to the following:
~username/AMS536-Spring2016/dock-tutorial/00.files/
/01.dockprep/
/02.surface-spheres/
/03.box-grid/
/04.dock/
/05.large-virtual-screen/
/06.virtual-screen/
/07.footprint/
/08.print_fps
|
In addition, most of the important files that are derived from the original crystal structure will be given a prefix that is the same as the PDB code, '3pgl'. The following sections in this tutorial will adhere to this directory structure/naming scheme.
II. Preparing the Receptor and Ligand
One issue that you will encounter when running Dock using the standard procedure on this system is that the ligand will move greatly. This is because normally all waters in the crystal structure are deleted, but they seem to be important for the positioning of the ligand. That causes the procedure in this tutorial to be slightly different from previous tutorials.
Download the PDB file (3PGL) and move file 3PGL.pdb to 00.files
3PGL.pdb was copied to raw_3pgl.pdb with command
cp 3PGL.pdb raw_3pgl.pdb
raw_3pgl.pdb was opened with VI terminal editor
Delete the header information, connect records, and waters except for waters 2, 21, 32, 39, and 258 (these numbers are listed in the 6th column of the dpb file). Change HETATM to ATOM, and delete all rows that have B in the fifth column Change RZX A to LIG B to make the ligand a different chain
Now open the system in chimera and use
Tools-->Structure Editing-->Add Charge Select AMBER ff99SB for Standard Residues, and add a charge of +2 to Mg and 0 to everything else
Prepare ligand and receptor files for dock
All waters were deleted from the file except for the ones believed to interact with the ligand and the Mg atom. These are believed to be important for the proper binding of the ligand. To properly orient the waters, run the system through tleap, keeping everything fixed except the 5 water molecules (this process can be seen in the amber tutorials on this wiki). This new system will yield better docking results, and should be saved as 3pgl.dockprep.mol2.
Open 3pgl.dockprep.mol2 in Chimera. Select the ligand and click Actions-->Atoms/Bonds-->delete. Save this file as 3pgl.rec.mol2.
With just the receptor open in Chimera, go to Select-->Chemistry-->element-->H and click Actions-->Atoms/Bonds-->delete. Save this file as 3pgl.rec.noH.mol2
Open 3pgl.dockprep.mol2 in Chimera and select the receptor. Click Actions-->Atoms/Bonds-->delete to get rid of everything except the ligand. Save this as 3pgl.lig.mol2.
III. Generating Receptor Surface and Spheres
Change directory to 02.surface-sphere Open 3pgl.rec.pdb in Chimera
To generate the molecular surface:
Action --> Surface --> Show
Save the .dms file
Tools --> Structure editing --> write DMS
.dms save to 3pgl.rec.dms
Create surface spheres
create input file INSPH
3pgl.rec.dms
R
X
0.0
4.0
1.4
3pgl.rec.sph
line 1 designates input file line 2 designates the generated spheres will be outside the receptor surface line 3 designates that all points on the receptor will be used line 4 designates the maximum surface radius of the spheres line 5 designates the minimum surface radius of the spheres line 6 designates the output file name
run sphgen
sphgen -i INSPH -o OUTSPH
sphgen is the sphere generation program from dock -i desginates the input file: INSPH -o designates the output file
At this point it is beneficial to visualize the spheres that were created. This can be done with chimera: Open chimera from the terminal choose file --> open 3pgl.rec.mol2 choose file --> open 3pgl.rec.sph
The image that appears should resemble this:
Then we need to select the spheres pertinent to our docking experiment. Usually these speheres will be the closest N spheres to the native ligand molecule.
Run
sphere_selector 3pgl.rec.sph ../01.dockprep/3pgl.lig.mol2 8.0
This command will select all of the spheres within 8.0 angstroms of the ligand and output them to selected_spheres.sph
To visualize the spheres using Chimera as previously done: Launch Chimera, choose File -> Open, choose 3pgl.rec.pdb File -> Open, choose output_spheres_selected.pdb Select -> Residue -> SPH Actions -> Atoms/Bonds -> sphere The selected spheres with the receptor surface should look similar to that as seen below:
IV. Generating Box and Grid
enter directory 03.box-grid
create input showbox.in
Y
8.0
../02.surface-spheres/selected_spheres.sph
1
3pgl.box.pdb
This input designates to dock that: we want to create a box the box length should be 8.0 Angstroms use the selected spheres in the file desginated output the box to the file specified
to use this input type:
showbox < showbox.in
This box can be visualized in chimera using a similar approach as visualizing the spheres
Compute the energy grid
create grid.in and grid out files using
touch grid.in
touch grid.out
Compute the grid, and save the output in the file grid.out
grid -i grid.in > grid.out
When prpmped, answer the questions as follows:
compute_grids yes grid_spacing 0.4 output_molecule no contact_score no energy_score yes energy_cutoff_distance 9999 atom_model a attractive_exponent 6 repulsive_exponent 9 distance_dielectric yes dielectric_factor 4 bump_filter yes bump_overlap 0.75 receptor_file ../01.dockprep/3pgl.rec.mol2 box_file ../03.box-grid/3pgl.box.pdb vdw_definition_file /opt/AMS536/dock6/parameters/vdw_AMBER_parm99.defn score_grid_prefix grid
this script should output verbosely to the terminal and produce 2 output files, grid.bmp and grid.nrg, both binary files.
V. Docking a Single Molecule for Pose Reproduction
Minimization
create min.in file and run dock with this as the input
touch min.in dock6 -i min.in
When prompted, answer the questions as follows:
conformer_search_type rigid use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ../01.dockprep/3pgl.lig.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ../01.dockprep/3pgl.lig.mol2 use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary no grid_score_secondary no multigrid_score_primary no multigrid_score_secondary no dock3.5_score_primary no dock3.5_score_secondary no continuous_score_primary yes continuous_score_secondary no cont_score_rec_filename ../01.dockprep/3pgl.rec.mol2 cont_score_att_exp 6 cont_score_rep_exp 12 cont_score_rep_rad_scale 1 cont_score_use_dist_dep_dielectric yes cont_score_dielectric 4.0 cont_score_vdw_scale 1 cont_score_es_scale 1 footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes simplex_max_iterations 1000 simplex_tors_premin_iterations 0 simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /opt/AMS536/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /opt/AMS536/dock6/parameters/flex.defn flex_drive_file /opt/AMS536/dock6/parameters/flex_drive.tbl ligand_outfile_prefix 3pgl.min write_orientations no num_scored_conformers 1 rank_ligands no
this command will output 3pgl.lig.min_scored.mol2
This structure can be visualized using chimera and loading the surface into chimera along with the minimized and unminimized ligand structures:
Footprint Calculation
At this point we are going to calculate the van der wals and electrostatic footprints for the unminimized and minimized ligand structures in relation to the receptor active site.
create the input file: footprint.in
conformer_search_type rigid use_internal_energy no ligand_atom_file ../3pgl.lig.min_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd no use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary no grid_score_secondary no multigrid_score_primary no multigrid_score_secondary no dock3.5_score_primary no dock3.5_score_secondary no continuous_score_primary no continuous_score_secondary no footprint_similarity_score_primary yes footprint_similarity_score_secondary no fps_use_footprint_reference_mol2 yes fps_footprint_reference_mol2_filename ../01.dockprep/3pgl.lig.mol2 fps_foot_compare_type Euclidean fps_normalize_foot no fps_foot_comp_all_residue yes fps_receptor_filename ../01.dockprep/3pgl.rec.mol2 fps_vdw_att_exp 6 fps_vdw_rep_exp 12 fps_vdw_rep_rad_scale 1 fps_use_distance_dependent_dielectric yes fps_dielectric 4.0 fps_vdw_fp_scale 1 fps_es_fp_scale 1 fps_hb_fp_scale 0 pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand no atom_model all vdw_defn_file /opt/AMS536/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /opt/AMS536/dock6/parameters/flex.defn flex_drive_file /opt/AMS536/dock6/parameters/flex_drive.tbl ligand_outfile_prefix fps.min.output write_footprints yes write_hbonds yes write_orientations no num_scored_conformers 1 rank_ligands no
this footprint calculation can be run in the same way as the minimization:
dock6 -i footprint.in
This will output three files: fps.min.output_scored.mol2
fps.min.output_scored_footprint_scored.txt
fps.min.output_hbond_scored.txt
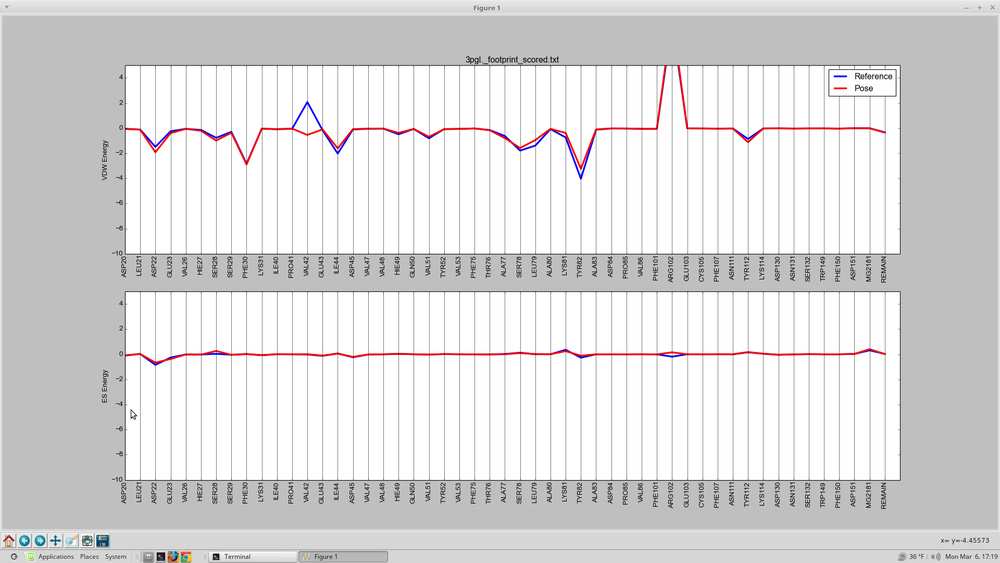
With an in house script, plot_footprint_single_magnitude.py the two sets of footprints will be plotted for comparison
this script will be run using:
python plot_footprint_single_magnitude.py fps.min.output_scored_footprint_scored.txt 50
The output will look something like this:
Rigid Docking
To run rigid docking, create rigid.in file and run dock with it as an input, use commands
touch rigid.in dock6 -i rigid.in
And answer the prompts as follows
conformer_search_type rigid use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ../01.dockprep/3pgl.lig.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ../01.dockprep/3pgl.lig.mol2 use_database_filter no orient_ligand yes automated_matching yes receptor_site_file ../02.surface-spheres/selected_spheres.sph max_orientations 1000 critical_points no chemical_matching no use_ligand_spheres no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ../03.box-grid/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes simplex_max_iterations 1000 simplex_tors_premin_iterations 0 simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_random_seed 0 simplex_restraint_min yes simplex_coefficient_restraint 10.0 atom_model all vdw_defn_file /opt/AMS536/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /opt/AMS536/dock6/parameters/flex.defn flex_drive_file /opt/AMS536/dock6/parameters/flex_drive.tbl ligand_outfile_prefix 3pgl_rigid_restraint write_orientations no num_scored_conformers 40 write_conformations no cluster_conformations yes cluster_rmsd_threshold 2.0 rank_ligands no
The output file is 3pgl.rigid_restraint_scored.mol2, and is saved in the directory from where you ran dock.
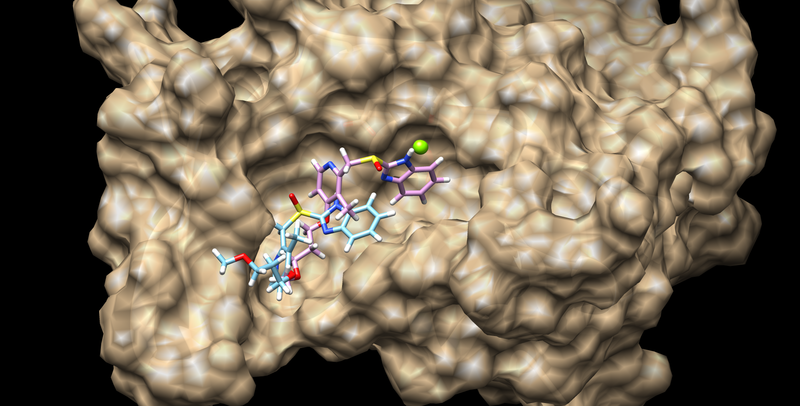
To vies the results, you can open the files of the receptor and ligand in CHIMERA from the 01.dockprep directory. The open your rigid docking results using viewdock:
Tools --> Surface/Binding Analysis --> ViewDock
Select the output file and set the file type to Dock 4,5,or 6. This should allow you to view the docked conformers ranked by grid score
The best scoring result should look like
Flexible Docking
To Run flexible docking, create flex.in and run dock6 with command
touch flex.in dock6 -i flex.in
When prompted, answer the questions as follows
conformer_search_type flex user_specified_anchor no limit_max_anchors no min_anchor_size 5 pruning_use_clustering yes pruning_max_orients 1000 pruning_clustering_cutoff 100 pruning_conformer_score_cutoff 100.0 pruning_conformer_score_scaling_factor 1.0 use_clash_overlap no write_growth_tree no use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ../01.dockprep/3pgl.lig.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ../3pgl.min_scored.mol2 use_database_filter no orient_ligand yes automated_matching yes receptor_site_file ../02.surface- spheres/selected_spheres.sph max_orientations 1000 critical_points no chemical_matching no use_ligand_spheres no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ../03.box-grid/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes minimize_anchor yes minimize_flexible_growth yes use_advanced_simplex_parameters no simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_anchor_max_iterations 500 simplex_grow_max_iterations 500 simplex_grow_tors_premin_iterations 0 simplex_random_seed 0 simplex_restraint_min yes simplex_coefficient_restraint 10.0 atom_model all vdw_defn_file /opt/AMS536/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /opt/AMS536/dock6/parameters/flex.defn flex_drive_file /opt/AMS536/dock6/parameters/flex_drive.tbl ligand_outfile_prefix 3pgl_flex write_orientations no num_scored_conformers 40 write_conformations no cluster_conformations yes cluster_rmsd_threshold 2.0 rank_ligands no
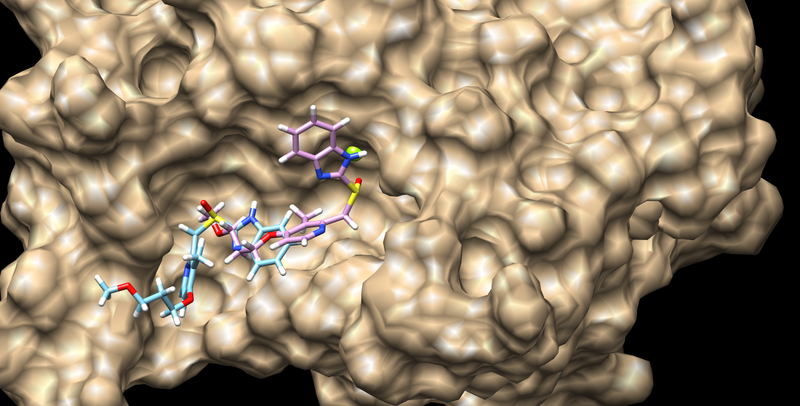
The output file is 3pgl.flex_scored.mol2, and is saved in the directory from where you ran dock.
These can be viewed in the same way as before and the best scoring result should look like
Fixed Anchor Docking
Along the same line as above, use
touch fad.in dock6 -i fad.in
When prompted, answer the questions as follows
Fix This
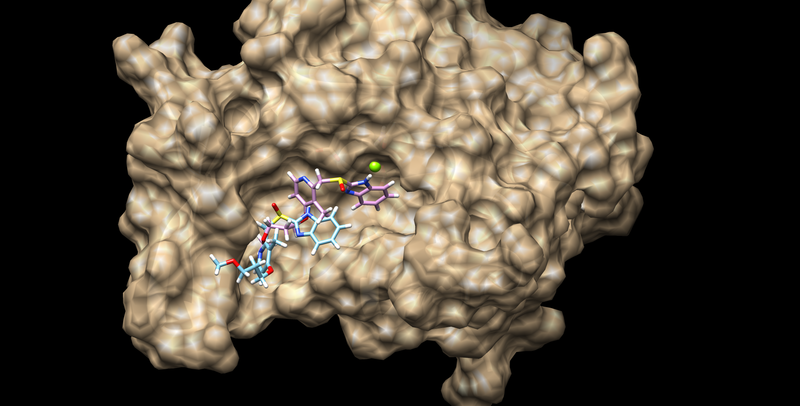
conformer_search_type flex user_specified_anchor no limit_max_anchors no min_anchor_size 5 pruning_use_clustering yes pruning_max_orients 1000 pruning_clustering_cutoff 100 pruning_conformer_score_cutoff 100.0 pruning_conformer_score_scaling_factor 1.0 use_clash_overlap no write_growth_tree no use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ../01.dockprep/3pgl.lig.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ../3pgl.min_scored.mol2 use_database_filter no orient_ligand yes automated_matching yes receptor_site_file ../02.surface- spheres/selected_spheres.sph max_orientations 1000 critical_points no chemical_matching no use_ligand_spheres no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ../03.box-grid/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes minimize_anchor yes minimize_flexible_growth yes use_advanced_simplex_parameters no simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_anchor_max_iterations 500 simplex_grow_max_iterations 500 simplex_grow_tors_premin_iterations 0 simplex_random_seed 0 simplex_restraint_min yes simplex_coefficient_restraint 10.0 atom_model all vdw_defn_file /opt/AMS536/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /opt/AMS536/dock6/parameters/flex.defn flex_drive_file /opt/AMS536/dock6/parameters/flex_drive.tbl ligand_outfile_prefix 3pgl_flex write_orientations no num_scored_conformers 40 write_conformations no cluster_conformations yes cluster_rmsd_threshold 2.0 rank_ligands no
The top scoring molecule is pictured below
VI. Virtual Screening
At this point we will begin docking potential inhibitors into the active site of 4qmz. The compound library we will use contains 25,000 molecules and was previously prepared for another docking experiment. The compound library contains molecules with rotatable bonds number between 7 and 15. The library used is titled small_ligand_library.mol2, this library can be found at /gpfs/projects/AMS536/virtual_screen_library on seawulf.
We began by copying all of our local directories outlined in the first section to a project directory on seawulf to allow use to expedite the docking process over the 25,000 compounds in our test library.
We will create a virtual-screen.in file that looks like the following:
conformer_search_type flex user_specified_anchor no limit_max_anchors no min_anchor_size 5 pruning_use_clustering yes pruning_max_orients 1000 pruning_clustering_cutoff 100 pruning_conformer_score_cutoff 100.0 pruning_conformer_score_scaling_factor 1.0 use_clash_overlap no write_growth_tree no use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file small_ligand_library.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ../01.dockprep/4qmz.lig.mol2 use_database_filter no orient_ligand yes automated_matching yes receptor_site_file ../02.surface-spheres/selected_spheres.sph max_orientations 1000 critical_points no chemical_matching no use_ligand_spheres no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ../03.box-grid/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes minimize_anchor yes minimize_flexible_growth yes use_advanced_simplex_parameters no simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_anchor_max_iterations 500 simplex_grow_max_iterations 500 simplex_grow_tors_premin_iterations 0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex_drive.tbl ligand_outfile_prefix 4qmz.virtualscreen write_orientations no num_scored_conformers 1 write_conformations no cluster_conformations yes cluster_rmsd_threshold 2.0 rank_ligands no
This script is submitted to the seawulf cluster using a script called screen.sh which contains the following:
#! /bin/tcsh #PBS -l nodes=4:ppn=28 #PBS -l walltime=24:00:00 #PBS -o jobstatus.out #PBS -e joberror.err #PBS -N large_vs #PBS -V #PBS -q long cd $PBS_O_WORKDIR mpirun -np 112 dock6.mpi -i virtual-screen.in -o virtualscreen1.out
This screen.sh input specifies that we will use 112 processors to dock our virtual library. We have a wall clock for the job of 24 hours and we will output jobstatus.out, joberror.err and virtualscreen1.out. In addition to these output files there is an output for the work performed by each processor individually.
Cartesian Minimization of the docked molecules
Now we will do cartesian minimization of the docked molecules. Cartesian here is referring to the receptor being explicitly present during the minimization.
Write the minimization input script minimize-vs.in:
conformer_search_type rigid use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file 4qmz.virtualscreen_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd no use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary no grid_score_secondary no multigrid_score_primary no multigrid_score_secondary no dock3.5_score_primary no dock3.5_score_secondary no continuous_score_primary yes continuous_score_secondary no cont_score_rec_filename ../01.dockprep/4qmz.rec.mol2 cont_score_att_exp 6 cont_score_rep_exp 12 cont_score_rep_rad_scale 1 cont_score_use_dist_dep_dielectric yes cont_score_dielectric 4.0 cont_score_vdw_scale 1 cont_score_es_scale 1 footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes simplex_max_iterations 1000 simplex_tors_premin_iterations 0 simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/flex_drive.tbl ligand_outfile_prefix 4qmz.virtualscreen1.minimized write_orientations no num_scored_conformers 1 rank_ligands no
Submit the minimization job using with the script "job-min.sh" following lines:
#!/bin/tcsh #PBS -l nodes=16:ppn=28 #PBS -l walltime=12:00:00 #PBS -o jobstatusmin.out #PBS -e jobstatusmin.err #PBS -N chunk0min #PBS -V #PBS -q medium module load intel/mpi/64/2017/0.098 cd $PBS_O_WORKDIR mpirun -np 448 /gpfs/projects/AMS536/zzz.programs/dock6/bin/dock6.mpi -i minimize-vs.in -o minimize-vs.out
At this point it would be good to rank our docked ligands by score and extract the 100 best scoring (100 lowest scoring, most negative) ligands to investigate further.