Difference between revisions of "2020 DOCK tutorial 1 with PDBID 3VJK"
Stonybrook (talk | contribs) (→Ligand Preparation) |
Stonybrook (talk | contribs) (→Rescoring Molecules) |
||
| (97 intermediate revisions by the same user not shown) | |||
| Line 8: | Line 8: | ||
'''3VJK''' is the PDB code for the crystal structure of human dipeptidyl peptidase IV, also known as DPP-4, with MP-513, which is called Teneligliptin [https://www.rcsb.org/structure/3vjk]. DPP-4 is a symmetrical dimer and has 729 residues per chain. The crystal has a resolution of 2.49 Å, a R-value of 0.279, and a R-free value of 0.225. To add on, the molecule in the crystal--Teneligliptin--has been approved for the treatment of type II Diabetes in Japan and has shown promising results in vivo [https://www.sciencedirect.com/science/article/pii/S0968089612006347?via%3Dihub]. | '''3VJK''' is the PDB code for the crystal structure of human dipeptidyl peptidase IV, also known as DPP-4, with MP-513, which is called Teneligliptin [https://www.rcsb.org/structure/3vjk]. DPP-4 is a symmetrical dimer and has 729 residues per chain. The crystal has a resolution of 2.49 Å, a R-value of 0.279, and a R-free value of 0.225. To add on, the molecule in the crystal--Teneligliptin--has been approved for the treatment of type II Diabetes in Japan and has shown promising results in vivo [https://www.sciencedirect.com/science/article/pii/S0968089612006347?via%3Dihub]. | ||
| − | == =Software packages === | + | ===Software packages === |
To follow this tutorial you will need to have the following programs installed: | To follow this tutorial you will need to have the following programs installed: | ||
| Line 18: | Line 18: | ||
At several points this tutorial will reference these programs as commands in a shell environment. The students who did this ran their programs on a UNIX (CoreOS or Ubuntu) server, although this process should generalize to your specific setup. For help, please reference available documentation. | At several points this tutorial will reference these programs as commands in a shell environment. The students who did this ran their programs on a UNIX (CoreOS or Ubuntu) server, although this process should generalize to your specific setup. For help, please reference available documentation. | ||
| − | |||
| − | |||
| − | |||
| − | |||
= Directory Organization = | = Directory Organization = | ||
| Line 51: | Line 47: | ||
File -> Open -> (Location where you downloaded PDB file) | File -> Open -> (Location where you downloaded PDB file) | ||
| − | [[File: | + | [[File:3vjk_pdb.png|thumb|center|800px]] |
The protein should appear the same as the image above. The image can be rotated to view from different angles. This is called a Ribbon diagram and shows the backbone of the protein, however some amino acid side chains are shown by default. Also shown explicitly are NAG amino acid modifications, the Oxygen of several water molecules and M51 (the ligand that is complexed with the protein). There are no Hydrogen atoms represented anywhere. This is because PDB files do not contain information on Hydrogen atoms. | The protein should appear the same as the image above. The image can be rotated to view from different angles. This is called a Ribbon diagram and shows the backbone of the protein, however some amino acid side chains are shown by default. Also shown explicitly are NAG amino acid modifications, the Oxygen of several water molecules and M51 (the ligand that is complexed with the protein). There are no Hydrogen atoms represented anywhere. This is because PDB files do not contain information on Hydrogen atoms. | ||
| Line 93: | Line 89: | ||
== Ligand Preparation== | == Ligand Preparation== | ||
| + | '''Preparing Ligand''' | ||
We will now need to prepare the Ligand, M-513. In a similar manner to receptor preparation, open the PDB file on Chimera. Likewise, you will also need to delete Chain B as previously stated. Now, you will be able to isolate the ligand by doing the following: | We will now need to prepare the Ligand, M-513. In a similar manner to receptor preparation, open the PDB file on Chimera. Likewise, you will also need to delete Chain B as previously stated. Now, you will be able to isolate the ligand by doing the following: | ||
select->residue->M-51 | select->residue->M-51 | ||
| Line 98: | Line 95: | ||
Actions->Atoms/Bonds->Delete | Actions->Atoms/Bonds->Delete | ||
You should be left with the following: | You should be left with the following: | ||
| − | [ | + | [[File:Ligand_RAW.png|thumb|center|800px|This figure shows what should be left after deleting everything but the ligand. This should show no hydrogens.]] |
| + | Next, we will save this as a mol2 file: | ||
| + | File->save as mol2 ->3VJK_ligand_noH.mol2 | ||
| + | '''Add Hydrogens and Charge''' | ||
| + | The crystal structure does not have any hydrogens because of technical limitations; hydrogen electron densities are too small to be detected. Consequently, we must add hydrogens to the ligand. | ||
| + | Tools->Structure editing-> add H | ||
| + | [[File:Ligand_with_H.png|thumb|center|800px|this figure shows how the ligand will appear once hydrogen are added]] | ||
| + | In a similar fashion, DOCK will need charges to perform calculations. | ||
| + | Tools->Structure editing-> Add Charge | ||
| + | It is important to make a note about the net charge of the ligand. '''you should not assume that chimera has the correct charge''''. You should look at the ligand and attempt to validate the charge, which should be '''+2''. | ||
| + | You can now save this as a mol2 file and name it: 3VJK_ligand_with_H.mol2 | ||
= Surface Generation & Sphere Selection = | = Surface Generation & Sphere Selection = | ||
| Line 112: | Line 119: | ||
Move this to the directory "002.surface_spheres" | Move this to the directory "002.surface_spheres" | ||
| − | [[File: | + | [[File:3vjk_protein.png|thumb|center|800px]] |
| Line 149: | Line 156: | ||
| − | [[File:sphgen_3vjk.png|thumb|center|500px|3vjk selected sphere -- | + | [[File:sphgen_3vjk.png|thumb|center|500px|3vjk selected sphere -- The spheres represent empty space in the protein structure]] |
| + | |||
| + | |||
| + | |||
| + | '''Sphere Selection''' | ||
| + | |||
| + | Using dock's sphere_selector script, we are able to produce a subset of spheres that are close (within 10 angstroms) to the ligand | ||
| + | |||
| + | sphere_selector 3vjk_receptor.sph 3vjk_ligand_H.mol2 10.0 | ||
| + | |||
| + | |||
| + | [[File:sphere_3vjk.png|thumb|center|500px|3vjk receptor with desired empty spaces highlighted]] | ||
| + | |||
| + | =Generation of the Box and Grid= | ||
| + | Energy calculations can be computationally expensive. Consequently, steps must be performed in order to reduce the number of calculations that are performed. In more detail, DOCK will be calculating the energy using a grid. We will be generating the grid; anything that is beyond the grid generated will not be in the calculation. This means that will ignore long distant interactions with ligand. | ||
| + | |||
| + | To start, we will be making a directory for the grid and the box | ||
| + | mkdir 003.gridbox | ||
| + | ==='''Generating the box'''=== | ||
| + | Next, we will be creating an input file that contains information for the Showbox programs. This file will contain parameters for the box. | ||
| + | vi showbox.in | ||
| + | we will put the following in to the file: | ||
| + | Y #generate box# | ||
| + | 8.0 #how many angstroms the box edges should be from the spheres# | ||
| + | ./../002.surface_spheres/select_spheres.sph #the location of the selected spheres# | ||
| + | 1 | ||
| + | 3VJK.box.pdb #name of the output file# | ||
| + | In order to run this you do this: | ||
| + | showbox < showbox.in | ||
| + | After you run this command a file called 3VJK.box.pdb will be generated. This files contains the grid. And can be visualized. | ||
| + | ==='''Generating the grid'''=== | ||
| + | In a similar manner, we will have to generate the grid. In order to do this we will need to make the input file for the grid program that contains: | ||
| + | compute_grids yes | ||
| + | grid_spacing 0.4 | ||
| + | output_molecule no | ||
| + | contact_score no | ||
| + | energy_score yes | ||
| + | energy_cutoff_distance 9999 | ||
| + | atom_model a | ||
| + | attractive_exponent 6 | ||
| + | repulsive_exponent 9 | ||
| + | distance_dielectric yes | ||
| + | dielectric_factor 4. | ||
| + | bump_filter yes | ||
| + | bump_overlap 0.75 | ||
| + | receptor_file d | ||
| + | box_file d.box.pbd | ||
| + | vdw_definition_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | score_grid_prefix grid | ||
| + | we will called this file grid.in. In order to generate the grid do the following: | ||
| + | grid -i grid.in -o gridinfo.out | ||
| + | the "-o" flag is used to specify the name of the output file. Once the program is completed there should be three files generated: gridinfo.out, grid.nrg, and grid.bmp. It is a good idea to make sure that gridinfo.out matches with the known information of the system. In other words, this is a good spot to double check your work. | ||
| + | =Energy Minimization= | ||
| + | Before running any dock calculations, we must take a moment to minimize the ligand. This is important because the current state of the ligand may not be at its lowest energy. We must take into consideration that crystallization can result in packing and other discrepancies that can impact our results. By minimizing the structure, we can make sure that none of the byproducts of crystalization will impact the results of the calculation. | ||
| + | |||
| + | We will make a new directory for Energy Minimization. | ||
| + | mkdir 004.energy_min | ||
| + | We will move into this directory. | ||
| + | Now, we can conduct the first step to conducting energy minimization is to create an input file. We will call this file min.in: | ||
| + | |||
| + | conformer_search_type rigid | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file ./../001.build/3VJK_ligand_hydrogens.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd yes | ||
| + | use_rmsd_reference_mol yes | ||
| + | rmsd_reference_filename ./../001.build/3VJK_ligand_hydrogens.mol2 | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix ./../003.gridbox/grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | simplex_max_iterations 1000 | ||
| + | simplex_tors_premin_iterations 0 | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_random_seed 0 | ||
| + | |||
| + | simplex_restraint_min yes | ||
| + | simplex_coefficient_restraint 10.0 | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix 3VJK.lig.min | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | Now that our input file is made we can now start running minimization. | ||
| + | dock6 -i min.in -o min.out | ||
| + | once this command is run two files will be generated: min.out and 3VJK.lig.min.mol2. The mol2 file that is generated can be visualized on chimera. | ||
| + | |||
| + | [[File:Minimized_ligand.png|thumb|center|800px|This figure shows the minimized ligand on top of the original ligand. There are some slight differences between the two structures.]] | ||
| + | |||
| + | =Short Cut Using Bash Scripting = | ||
| + | Running the previous steps can become tedious when one is working with a massive set of systems. Moreover, the use of a bash script can help to keep a record of what parameters were used for an experiment. A quick way to run these steps is with the following script: | ||
| + | #!/bin/sh | ||
| + | echo PDB name | ||
| + | read pdb | ||
| + | echo receptor file with hydrogen | ||
| + | read receptor | ||
| + | echo receptor file DMA | ||
| + | read receptor_DMS | ||
| + | echo ligand file | ||
| + | read ligand | ||
| + | EOF | ||
| + | echo generating spheres | ||
| + | cat > INSPH << EOF | ||
| + | $receptor_DMS | ||
| + | R | ||
| + | X | ||
| + | 0.0 | ||
| + | 4.0 | ||
| + | 1.4 | ||
| + | ${pdb}_receptor.sph | ||
| + | EOF | ||
| + | echo selecting spheres | ||
| + | sphgen -i INSPH -o OUTSPH | ||
| + | sphere_selector pdb_receptor.sph $ligand 10.0 | ||
| + | #generate grid | ||
| + | echo generating grid | ||
| + | cat > showbox.in << EOF | ||
| + | Y | ||
| + | 8.0 | ||
| + | ${pdb}_receptor.sph | ||
| + | 1 | ||
| + | ${pdb}.box.pdb | ||
| + | EOF | ||
| + | showbox<showbox.in | ||
| + | cat > grid.in << EOF | ||
| + | compute_grids yes | ||
| + | grid_spacing 0.4 | ||
| + | output_molecule no | ||
| + | contact_score no | ||
| + | energy_score yes | ||
| + | energy_cutoff_distance 9999 | ||
| + | atom_model a | ||
| + | attractive_exponent 6 | ||
| + | repulsive_exponent 9 | ||
| + | distance_dielectric yes | ||
| + | dielectric_factor 4 | ||
| + | bump_filter yes | ||
| + | bump_overlap 0.75 | ||
| + | receptor_file $receptor | ||
| + | box_file pdb.box.pdb | ||
| + | vdw_definition_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | score_grid_prefix grid | ||
| + | EOF | ||
| + | grid -i grid.in -o gridinfo.out | ||
| + | cat > min.in << EOF | ||
| + | conformer_search_type rigid | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | enternal_energy_cutoff 100.0 | ||
| + | ligand_atom_file $ligand | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd yes | ||
| + | use_rmsd_reference_mol yes | ||
| + | rmsd_reference_filename $ligand | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | simplex_max_iterations 1000 | ||
| + | simplex_tors_premin_iterations 0 | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min yes | ||
| + | simplex_coefficient_restraint 10.0 | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix pdb.lig.min | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | EOF | ||
| + | echo minimized | ||
| + | dock6 -i min.in -o min.out | ||
| + | This can all be copied into a document named dock_setup.sh. This can become an executable file by using the command: | ||
| + | chmod u+x dock_setup.sh | ||
| + | |||
| + | =Footprint Analysis= | ||
| + | Using the electrostatic interactions and Van Der Waals Interactions, the molecular footprint can be used to understand how the ligand binds the receptor. Residues that are more negative are predominantly involved in the interaction. | ||
| + | |||
| + | First we will create a directory for the molecular footprint | ||
| + | mkdir 005.footprint | ||
| + | In this space, we will be creating the files that will be used for the molecular footprint. The first thing we have to do is make the footprint.in file: | ||
| + | conformer_search_type rigid | ||
| + | use_internal_energy no | ||
| + | ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd no | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary no | ||
| + | grid_score_secondary no | ||
| + | multigrid_score_primary no | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_primary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_primary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_primary yes | ||
| + | footprint_similarity_score_secondary no | ||
| + | fps_score_use_footprint_reference_mol2 yes | ||
| + | fps_score_footprint_reference_mol2_filename ./../001.build/3VJK_ligand_hydrogens.mol2 | ||
| + | fps_score_foot_compare_type Euclidean | ||
| + | fps_score_normalize_foot no | ||
| + | fps_score_foot_comp_all_residue yes | ||
| + | fps_score_receptor_filename ./../001.build/3VJK_hydrogen_protein.mol2 | ||
| + | fps_score_vdw_att_exp 6 | ||
| + | fps_score_vdw_rep_exp 9 | ||
| + | fps_score_vdw_rep_rad_scale 1 | ||
| + | fps_score_use_distance_dependent_dielectric yes | ||
| + | fps_score_dielectric 4.0 | ||
| + | fps_score_vdw_fp_scale 1 | ||
| + | fps_score_es_fp_scale 1 | ||
| + | fps_score_hb_fp_scale 0 | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix footprint.out | ||
| + | write_footprints yes | ||
| + | write_hbonds yes | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | Now you can use the following command to make the footprint: | ||
| + | dock6 -i footprint.in | ||
| + | Once this is completed you should have three new files: footprint.out_footprint_scored.txt footprint.out_hbond_scored.txt and footprint.out_scored.mol2 | ||
| + | |||
| + | We can visualize the results using a python script: /gpfs/projects/AMS536/zzz.programs/plot_footprint_single_magnitude.py . This should be copied to the current directory. | ||
| + | cp /gpfs/projects/AMS536/2020/536_class/steve_ta/footprint_test_1.21.2020/plot_footprint_single_magnitude.py | ||
| + | |||
| + | This command can run by doing the following: | ||
| + | python plot_footprint_single_magnitude.py footprint.out_footprint_scored.txt 50 | ||
| + | This will show the energetic distribution for the 50 most significant residues. | ||
| + | |||
| + | [[File:3vjkfootprint.png|center]] | ||
| + | |||
| + | This distribution shows the relative stability of each residue with respect to the ligand before and after the docking process. In general, the docking process should arrange the ligand such that the global stability of the compound has increased. | ||
| + | |||
| + | =Docking= | ||
| + | ===Rigid Docking=== | ||
| + | This is the least computationally expensive docking method. This docking routine does not sample internal degrees of freedom (bond angles). The ligand is treated as a rigid object which is why minimization was performed in the prior step. | ||
| + | |||
| + | The first step is to create a dictory--where we will be putting are input and output files. | ||
| + | mkdir 006.rigid_docking | ||
| + | Now that we have the directory ready, we can make documents within this. The first thing we will want to do is set up the input file | ||
| + | touch rigid.in | ||
| + | dock6 -i rigid.in | ||
| + | This will bring up an interactive input screen. Unlike the prior sets, it is highly suggested to manually complete this task because some options may be moved around depending on the version used. Below you can find the parameters that were used for this tutorial. | ||
| + | conformer_search_type flex | ||
| + | write_fragment_libraries no | ||
| + | user_specified_anchor no | ||
| + | limit_max_anchors no | ||
| + | min_anchor_size 5 | ||
| + | pruning_use_clustering yes | ||
| + | pruning_max_orients 1000 | ||
| + | pruning_clustering_cutoff 100 | ||
| + | pruning_conformer_score_cutoff 100.0 | ||
| + | pruning_conformer_score_scaling_factor 1.0 | ||
| + | use_clash_overlap no | ||
| + | write_growth_tree no | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd yes | ||
| + | use_rmsd_reference_mol yes | ||
| + | rmsd_reference_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | use_database_filter no | ||
| + | orient_ligand yes | ||
| + | automated_matching yes | ||
| + | receptor_site_file ./../002.surface_spheres/selected_spheres.sph | ||
| + | max_orientations 1000 | ||
| + | critical_points no | ||
| + | chemical_matching no | ||
| + | use_ligand_spheres no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix ./../003.gridbox/grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | minimize_anchor yes | ||
| + | minimize_flexible_growth yes | ||
| + | use_advanced_simplex_parameters no | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_anchor_max_iterations 500 | ||
| + | simplex_grow_max_iterations 500 | ||
| + | simplex_grow_tors_premin_iterations 0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix rigid.out | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | Once the input is complete, there should be two output files: rigid.out and rigid.out_scored.mol2. Like the previous set with minimization, the results can be seen with chimera. | ||
| + | [[File:Rigid_entropy.png|thumb|center|800px|the figure shows the protein with the minimized ligand and the ligand from the rigid dock experiment]] | ||
| + | |||
| + | ===Fixed Anchor Docking=== | ||
| + | In this type of docking, the largest molecular fragment (the anchor) is oriented as the first step. The anchor is defined as the moiety containing no internal rotatable bonds possessing the most atoms. Subsequent rigid portions are attached and are oriented to be as energetically favorable as possible. This allows a larger space of sampling, but is still confined. | ||
| + | |||
| + | Make a directory called 007.fixed_anchor_docking/ | ||
| + | |||
| + | In this directory, we will be creating an input file (like we did for the rigid docking) that is called flex.in | ||
| + | conformer_search_type flex | ||
| + | write_fragment_libraries no | ||
| + | user_specified_anchor no | ||
| + | limit_max_anchors no | ||
| + | min_anchor_size 5 | ||
| + | pruning_use_clustering yes | ||
| + | pruning_max_orients 1000 | ||
| + | pruning_clustering_cutoff 100 | ||
| + | pruning_conformer_score_cutoff 100.0 | ||
| + | pruning_conformer_score_scaling_factor 1.0 | ||
| + | use_clash_overlap no | ||
| + | write_growth_tree no | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd yes | ||
| + | use_rmsd_reference_mol yes | ||
| + | rmsd_reference_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix ./../003.gridbox/grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | minimize_anchor yes | ||
| + | minimize_flexible_growth yes | ||
| + | use_advanced_simplex_parameters no | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge .1 | ||
| + | simplex_cycle_converge 1 | ||
| + | simplex_trans_step 1 | ||
| + | simplex_rot_step .1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_anchor_max_iterations 500 | ||
| + | simplex_grow_max_iterations 500 | ||
| + | simplex_grow_tors_premin_iterations 0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | This can be run by doing the following: | ||
| + | dock6 -i fixed.in -o fixed.out | ||
| + | Once this is completed you should have two output files: flex.out and 2vjk_fad_scored.mol2. | ||
| + | [[File:3VJK_FAD.png|thumb|center|800px|the figure shows the protein with the minimized ligand and the ligand from the fixed anchor dock experiment]] | ||
| + | |||
| + | ===Flexible Docking=== | ||
| + | Finally, this is the most expensive docking. This will sample all the internal degrees of freedom within the domain of bond angles. | ||
| + | |||
| + | Once again create a directory for this docking experiment. We will call it 008.flexible_docking | ||
| + | |||
| + | conformer_search_type flex | ||
| + | write_fragment_libraries no | ||
| + | user_specified_anchor no | ||
| + | limit_max_anchors no | ||
| + | min_anchor_size 5 | ||
| + | pruning_use_clustering yes | ||
| + | pruning_max_orients 1000 | ||
| + | pruning_clustering_cutoff 100 | ||
| + | pruning_conformer_score_cutoff 100.0 | ||
| + | pruning_conformer_score_scaling_factor 1.0 | ||
| + | use_clash_overlap no | ||
| + | write_growth_tree no | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd yes | ||
| + | use_rmsd_reference_mol yes | ||
| + | rmsd_reference_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | use_database_filter no | ||
| + | orient_ligand yes | ||
| + | automated_matching yes | ||
| + | receptor_site_file ./../002.surface_spheres/selected_spheres.sph | ||
| + | max_orientations 1000 | ||
| + | critical_points no | ||
| + | chemical_matching no | ||
| + | use_ligand_spheres no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix ./../003.gridbox/grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | minimize_anchor yes | ||
| + | minimize_flexible_growth yes | ||
| + | use_advanced_simplex_parameters no | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_anchor_max_iterations 500 | ||
| + | simplex_grow_max_iterations 500 | ||
| + | simplex_grow_tors_premin_iterations 0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix flex.out | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | |||
| + | Like the previous docking experiments we will do the following to make it run: | ||
| + | dock6 -i flex.in -o flex.out | ||
| + | Two files should have been generated: flex.out and flex.out_scored.mol2 | ||
| + | [[File:3VJK_FLEXIBLE.png|thumb|center|800px| the figure shows the protein with the minimized ligand and the ligand from the flexible dock experiment]] | ||
| + | |||
| + | = Virtual Screening = | ||
| + | |||
| + | Virtual screening is the process of passing a library of molecules through DOCK to a single receptor. This library of molecules can be tailored by the user to match the desired size and constituents. The advantage of a virtual screen is that many molecules can be analyzed with minimal user input, and the most strong binding molecules can be selected for further analysis. The screen performed for this tutorial used the library file "VS_library_5K.mol2" which can be found within the AMS536 directory. If desired to use a different library, all of the molecules must be provided within a single multi-mol2 file. | ||
| + | |||
| + | touch virtual.in | ||
| + | |||
| + | Input the following prompts: | ||
| + | |||
| + | conformer_search_type flex | ||
| + | write_fragment_libraries no | ||
| + | user_specified_anchor no | ||
| + | limit_max_anchors no | ||
| + | min_anchor_size 5 | ||
| + | pruning_use_clustering yes | ||
| + | pruning_max_orients 1000 | ||
| + | pruning_clustering_cutoff 100 | ||
| + | pruning_conformer_score_cutoff 100.0 | ||
| + | pruning_conformer_score_scaling_factor 1.0 | ||
| + | use_clash_overlap no | ||
| + | write_growth_tree no | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 9 | ||
| + | internal_energy_cutoff 100.0 | ||
| + | ligand_atom_file VS_library_5K.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd no | ||
| + | use_database_filter no | ||
| + | orient_ligand yes | ||
| + | automated_matching yes | ||
| + | receptor_site_file ../002.surface_spheres/selected_spheres.sph | ||
| + | max_orientations 1000 | ||
| + | critical_points no | ||
| + | chemical_matching no | ||
| + | use_ligand_spheres no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary yes | ||
| + | grid_score_secondary no | ||
| + | grid_score_rep_rad_scale 1 | ||
| + | grid_score_vdw_scale 1 | ||
| + | grid_score_es_scale 1 | ||
| + | grid_score_grid_prefix ../003.gridbox/grid | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | minimize_anchor yes | ||
| + | minimize_flexible_growth yes | ||
| + | use_advanced_simplex_parameters no | ||
| + | simplex_max_cycles 1 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_anchor_max_iterations 500 | ||
| + | simplex_grow_max_iterations 500 | ||
| + | simplex_grow_tors_premin_iterations 0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix virtual.out | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | |||
| + | Thus far all work has likely been done on a local machine. Running Dock on 5000 molecules could take an extended time if done on a local machine. To maximize the use of resources, a virtual screen can be done on the Seawolf Cluster. In order to do this, a submission script must be written which gives the cluster information on how to allocate resources. | ||
| + | |||
| + | Name the submission script virtual_screen.sh | ||
| + | |||
| + | vi virtual_screen.sh | ||
| + | |||
| + | #!/bin/sh | ||
| + | #SBATCH --partition=long-40core | ||
| + | #SBATCH --time=48:00:00 | ||
| + | #SBATCH --nodes=1 | ||
| + | #SBATCH --ntasks=1 | ||
| + | #SBATCH --job-name=536.vs | ||
| + | #SBATCH --output=536.vs.out | ||
| + | |||
| + | dock6 -i virtual.in -o virtual.out | ||
| + | |||
| + | To submit the script in the bash shell: | ||
| + | sbatch virtual_screen.sh | ||
| + | You may check the status of you job: | ||
| + | squeue -u {username} | ||
| + | Once the job is completed you should read the output files. | ||
| + | cat 536.vs.out | ||
| + | Since only one node and 1 core, it is expected that the job was canceled due to reaching the time limit. | ||
| + | [[File:Time_limit.png|thumb|center|800px|the image shows the error that will most likely occur when a virtual screening of 5k molecules is conducted on this system]] | ||
| + | |||
| + | We can also see how many molecules were processed using this command: | ||
| + | grep Molecule: virtual_screening.out| wc -l | ||
| + | You may also visualize the results of the virtual screening by downloading virtual.out_scored.mol2 and using chimera | ||
| + | Tools-> Surface/BindingAnalysis->View Dock-> Dock6 | ||
| + | This will allow you to visualize each molecule that was screened. Next, you can look at the properties of the molecules by downing the following: | ||
| + | Column->show | ||
| + | You can select from a variety of properties and rank the molecules based on these properties. This is useful for analyzing which molecules are favored in the active site | ||
| + | |||
| + | Consequently, we must re-run the job with a large number of nodes and cores using MPI. | ||
| + | |||
| + | =Virtual Screening with MPI= | ||
| + | In the previous section, it was found that the time limit was reached-- however--not all the molecules were screened. We simply cannot resubmit the job to complete the analysis. To compensate, we can submit the job to run on multiple cores. We can do this by using MPI. | ||
| + | |||
| + | You should first start off by making a new directory for the MPI Virtual Screening. This will be called 012.MPI_VS. You can simply copy the input file from the previous run. The only change will be in the submission script: | ||
| + | #!/bin/sh | ||
| + | #SBATCH --partition=long-40core | ||
| + | #SBATCH --time=48:00:00 | ||
| + | #SBATCH --nodes=4 | ||
| + | #SBATCH --ntasks=32 | ||
| + | #SBATCH --job-name=tutorial_run | ||
| + | #SBATCH --output=%x-%j.o | ||
| + | |||
| + | echo "starting DOCK6.9 Simulation" | ||
| + | module load intel/mpi/64/2018/18.0.3 | ||
| + | |||
| + | mpirun -np 32 dock6.mpi -i virtual_screening.in -o virtual_screening.out | ||
| + | echo "done" | ||
| + | In this script, I indicated that I will be using 4 nodes and a total of 32 cores. Additionally, it is required to load the module for mpi and use the dock6.mpi executable. | ||
| + | Once the job has finish running you should find that a major of the molecules were calculated. Once again you may check using this command: | ||
| + | grep Molecule: virtual_screening.out| wc -l | ||
| + | In this tutorial, the 32 cores allowed for the calculation of 5665 out of the 5730 small molecules in the library. Thus, in the future, it may be required to allocate more resources to run through the whole library. Nevertheless, we may proceed using the available data. | ||
| + | =Cartesian Minimization= | ||
| + | We will now conduct minimization on the molecules that were docked. To do this, we must first create an input file: | ||
| + | conformer_search_type rigid | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100 | ||
| + | ligand_atom_file ./../012.mpi_job/virtual.out_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd no | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary no | ||
| + | grid_score_secondary no | ||
| + | multigrid_score_primary no | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_primary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_primary yes | ||
| + | continuous_score_secondary no | ||
| + | cont_score_rec_filename ./../001.build/3VJK_hydrogen_protein.mol2 | ||
| + | cont_score_att_exp 6 | ||
| + | cont_score_rep_exp 12 | ||
| + | cont_score_rep_rad_scale 1 | ||
| + | cont_score_use_dist_dep_dielectric yes | ||
| + | cont_score_dielectric 4.0 | ||
| + | cont_score_vdw_scale 1.0 | ||
| + | cont_score_es_scale 1.0 | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_secondary no | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand yes | ||
| + | simplex_max_iterations 1000 | ||
| + | simplex_tors_premin_iterations 0 | ||
| + | simplex_max_cycles 1.0 | ||
| + | simplex_score_converge 0.1 | ||
| + | simplex_cycle_converge 1.0 | ||
| + | simplex_trans_step 1.0 | ||
| + | simplex_rot_step 0.1 | ||
| + | simplex_tors_step 10.0 | ||
| + | simplex_random_seed 0 | ||
| + | simplex_restraint_min no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | ligand_outfile_prefix 3vjk.virtual_screen.min | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | We have named this min.in. It is important to make sure that the directory paths reflect your own paths. Once the input file has been made you should create a submission script: | ||
| + | #!/bin/sh | ||
| + | #SBATCH --partition=long-40core | ||
| + | #SBATCH --time=48:00:00 | ||
| + | #SBATCH --nodes=3 | ||
| + | #SBATCH --ntasks=24 | ||
| + | #SBATCH --job-name=tutorial_run | ||
| + | #SBATCH --dependency=afterok:227294 | ||
| + | #SBATCH --output=%x-%j.o | ||
| + | |||
| + | echo "starting Cartesian Minimization" | ||
| + | dock6 -i min.in -o min.out | ||
| + | echo "Minimization done" | ||
| + | You may name this job.sh. And now you should be able to submit the job. | ||
| − | + | In a similar manner to the previous section, you can download the output .mol2 files and load it using viewdock on chimera. This can be used to compare the unminimized ligands to the minimized ligands. You should expect to find that the energy is more negative after minimization. | |
| + | [[File:3vjk_mpi_minimized.png|thumb|center|800px|the image shows the protein with both the minimized ligand and unminimized ligand. The minimized ligand is colored yellow]] | ||
| − | = | + | =Rescoring Molecules= |
| + | We will now rescore the molecules based on several factors. This includes footprint, pharmacophore, Tanimoto, Hungarian, and volume_overlap score. The first step is to make a directory. This can be called rescore. Now you must make the input file (rescore.in): | ||
| + | conformer_search_type rigid | ||
| + | use_internal_energy yes | ||
| + | internal_energy_rep_exp 12 | ||
| + | internal_energy_cutoff 100 | ||
| + | ligand_atom_file ./../009.virtual_screening/virtual.out_scored.mol2 | ||
| + | limit_max_ligands no | ||
| + | skip_molecule no | ||
| + | read_mol_solvation no | ||
| + | calculate_rmsd no | ||
| + | use_database_filter no | ||
| + | orient_ligand no | ||
| + | bump_filter no | ||
| + | score_molecules yes | ||
| + | contact_score_primary no | ||
| + | contact_score_secondary no | ||
| + | grid_score_primary no | ||
| + | grid_score_secondary no | ||
| + | multigrid_score_primary no | ||
| + | multigrid_score_secondary no | ||
| + | dock3.5_score_primary no | ||
| + | dock3.5_score_secondary no | ||
| + | continuous_score_primary no | ||
| + | continuous_score_secondary no | ||
| + | footprint_similarity_score_primary no | ||
| + | footprint_similarity_score_secondary no | ||
| + | pharmacophore_score_primary no | ||
| + | pharmacophore_score_secondary no | ||
| + | descriptor_score_primary yes | ||
| + | descriptor_score_secondary no | ||
| + | descriptor_use_grid_score no | ||
| + | descriptor_use_multigrid_score no | ||
| + | descriptor_use_continuous_score no | ||
| + | descriptor_use_footprint_similarity yes | ||
| + | descriptor_use_pharmacophore_score yes | ||
| + | descriptor_use_hungarian yes | ||
| + | descriptor_use_volume_overlap yes | ||
| + | descriptor_fps_score_use_footprint_reference_mol2 yes | ||
| + | descriptor_fps_score_footprint_reference_mol2_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | descriptor_fps_score_foot_compare_type Euclidean | ||
| + | descriptor_fps_score_normalize_foot no | ||
| + | descriptor_fps_score_foot_comp_all_residue yes | ||
| + | descriptor_fps_score_receptor_filename ./../001.build/3VJK_hydrogen_protein.mol2 | ||
| + | descriptor_fps_score_vdw_att_exp 6 | ||
| + | descriptor_fps_score_vdw_rep_exp 12 | ||
| + | descriptor_fps_score_vdw_rep_rad_scale 1 | ||
| + | descriptor_fps_score_use_distance_dependent_dielectric yes | ||
| + | descriptor_fps_score_dielectric 4.0 | ||
| + | descriptor_fps_score_vdw_fp_scale 1 | ||
| + | descriptor_fps_score_es_fp_scale 1 | ||
| + | descriptor_fps_score_hb_fp_scale 0 | ||
| + | descriptor_fms_score_use_ref_mol2 yes | ||
| + | descriptor_fms_score_ref_mol2_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | descriptor_fms_score_write_reference_pharmacophore_mol2 no | ||
| + | descriptor_fms_score_write_reference_pharmacophore_txt no | ||
| + | descriptor_fms_score_write_candidate_pharmacophore no | ||
| + | descriptor_fms_score_write_matched_pharmacophore no | ||
| + | descriptor_fms_score_compare_type overlap | ||
| + | descriptor_fms_score_full_match yes | ||
| + | descriptor_fms_score_match_rate_weight 5.0 | ||
| + | descriptor_fms_score_match_dist_cutoff 1.0 | ||
| + | descriptor_fms_score_match_proj_cutoff .7071 | ||
| + | descriptor_fms_score_max_score 20 | ||
| + | descriptor_fingerprint_ref_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | descriptor_hms_score_ref_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | descriptor_hms_score_matching_coeff -5 | ||
| + | descriptor_hms_score_rmsd_coeff 1 | ||
| + | descriptor_volume_score_reference_mol2_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 | ||
| + | descriptor_volume_score_overlap_compute_method analytical | ||
| + | descriptor_weight_fps_score 1 | ||
| + | descriptor_weight_pharmacophore_score 1 | ||
| + | descriptor_weight_fingerprint_tanimoto -1 | ||
| + | descriptor_weight_hms_score 1 | ||
| + | descriptor_weight_volume_overlap_score -1 | ||
| + | gbsa_zou_score_secondary no | ||
| + | gbsa_hawkins_score_secondary no | ||
| + | SASA_score_secondary no | ||
| + | amber_score_secondary no | ||
| + | minimize_ligand no | ||
| + | atom_model all | ||
| + | vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn | ||
| + | flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn | ||
| + | flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl | ||
| + | chem_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/chem.defn | ||
| + | pharmacophore_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/ph4.defn | ||
| + | ligand_outfile_prefix descriptor.output | ||
| + | write_footprints yes | ||
| + | write_hbonds yes | ||
| + | write_orientations no | ||
| + | num_scored_conformers 1 | ||
| + | rank_ligands no | ||
| + | In a similar fashion to submitting the virtual screening, you may chose to submit the job using script: | ||
| + | #!/bin/sh | ||
| + | #SBATCH --partition=long-40core | ||
| + | #SBATCH --time=48:00:00 | ||
| + | #SBATCH --nodes=4 | ||
| + | #SBATCH --ntasks=32 | ||
| + | #SBATCH --job-name=tutorial_run | ||
| + | #SBATCH --output=%x-%j.o | ||
| + | |||
| + | echo "starting DOCK6.9 Simulation" | ||
| + | module load intel/mpi/64/2018/18.0.3 | ||
| + | |||
| + | mpirun -np 32 dock6.mpi -i rescore.in -o rescore.out | ||
| + | echo "done" | ||
| + | This can be submitted by doing the following | ||
| + | sbatch mpi.sh | ||
| + | Once the job has run you should be able to see the results by loading descriptor.output_scored.mol2 into chimera using view docks. You can select which scoring properties to look at by using the column feature. | ||
Latest revision as of 00:23, 16 March 2020
Contents
- 1 Introduction
- 2 Directory Organization
- 3 Receptor Preparation
- 4 Surface Generation & Sphere Selection
- 5 Generation of the Box and Grid
- 6 Energy Minimization
- 7 Short Cut Using Bash Scripting
- 8 Footprint Analysis
- 9 Docking
- 10 Virtual Screening
- 11 Virtual Screening with MPI
- 12 Cartesian Minimization
- 13 Rescoring Molecules
Introduction
DOCK
DOCK 6 is a molecular modeling software that is used for investigating ligand binding geometry and ligand interactions[1]. Consequently, its relevance in the field of drug discovery is clear. This program was initially developed by Dr. Irwin Kuntz and colleagues at the University of California San Francisco. A major feature of DOCK 6 is the search algorithm that is used: “anchor-and-grow”; this sets the software apart from its counterparts [2]. This method first identifies the rigid structure of a particular ligand--anchors-- then the program docks the ligand using its geometry. Following the docking, a partial conformational search is performed. In simple terms, the positions of the anchor are allowed independently however once a favored conformation is found it is retained. Once this step is completed, energy minimization is carried out Full details here.
3VJK
3VJK is the PDB code for the crystal structure of human dipeptidyl peptidase IV, also known as DPP-4, with MP-513, which is called Teneligliptin [3]. DPP-4 is a symmetrical dimer and has 729 residues per chain. The crystal has a resolution of 2.49 Å, a R-value of 0.279, and a R-free value of 0.225. To add on, the molecule in the crystal--Teneligliptin--has been approved for the treatment of type II Diabetes in Japan and has shown promising results in vivo [4].
Software packages
To follow this tutorial you will need to have the following programs installed:
- DOCK
- Chimera
This tutorial used Dock 6.9 & Chimera 1.13.1
At several points this tutorial will reference these programs as commands in a shell environment. The students who did this ran their programs on a UNIX (CoreOS or Ubuntu) server, although this process should generalize to your specific setup. For help, please reference available documentation.
Directory Organization
The following tutorial will use the organization of directories prepared below. This specific organization is not required but is recommended. The "mkdir" command will be employed which creates a new folder in which files can be saved. To navigate into a directory use the command "cd" followed by the directory name. To change to the directory the next level up, use the command "cd .." .
Within the Bash Shell environment:
mkdir 3VJK cd 3VJK mkdir 001.structure 002.surface_spheres 003.gridbox 004.dock 005.virtual_screen 006.virtual_screen_mpi 007.cartesianmin 008.rescore
All eight directories should be created now and this can be visually confirmed with the command "ls".
Receptor Preparation
Preparing the Structure for Docking
Downloading and Opening PDB File
Download the PDB Format file from the associated rcsb page here. This web page includes associated articles, files, and other meta data.
Download files -> PDB Format
This file provides information on the 3D orientation of the atoms within the protein and ligand as well as any co-factors (any other molecules present during the crystallization experiment, typically water and metal ions). The file can be opened up and manipulated in the program Chimera.
Open Chimera:
File -> Open -> (Location where you downloaded PDB file)
The protein should appear the same as the image above. The image can be rotated to view from different angles. This is called a Ribbon diagram and shows the backbone of the protein, however some amino acid side chains are shown by default. Also shown explicitly are NAG amino acid modifications, the Oxygen of several water molecules and M51 (the ligand that is complexed with the protein). There are no Hydrogen atoms represented anywhere. This is because PDB files do not contain information on Hydrogen atoms.
Preparation of the Protein Receptor for Docking
Docking requires that the protein receptor and ligand be separated into different files. First, the receptor file will be prepared. This particular protein is a homo-dimer (two identical units of the same peptide). For simplicity and to avoid possible complications in later steps, only one of the peptide chains will be retained. This step should be applied judiciously in protein systems where the ligand is at the interface of two dimers.
Select -> Chain -> B Actions -> Atoms/ Bonds -> Delete
Only one monomer of protein should remain now.
Next the NAG amino acid modifications, waters and ligand will be removed. They are not crucial for the Docking experiment, and may be problematic and cause failure if retained.
Select -> Residue -> All nonstandard Actions -> Atoms/ Bonds -> Delete
The receptor is now "clean" and should be saved prior to the next step.
File -> Save Mol2 -> "3VJK_rec_woH.mol2"
It is important to give files a logical naming scheme. The woH portion is to specify Hydrogens have not yet been added. Move this file to the directory "001.structure"
Adding Hydrogens and Charge
In order to calculate interactions between the protein and ligand, Hydrogens must be added to the receptor. Chimera will apply standard protonation states to the amino acids. It is important to check these protonation states afterwards, as they may not match the crystallization experiment. For example, the paper associated with the PDB being worked with may specify a certain residue is protonated. It would then be crucial to check this after the following step, and if it is incorrect, to adjust it manually.
Structure Editing -> Add H -> Ok
Next partial charges will be added to each atom in the receptor.
Structure Editing -> Add Charge -> (AM1BCC charges should be selected) -> Ok
Now save this as a mol2 file "3VJK_rec_dockprep.mol2" and move it to the directory "001.structure"
Ligand Preparation
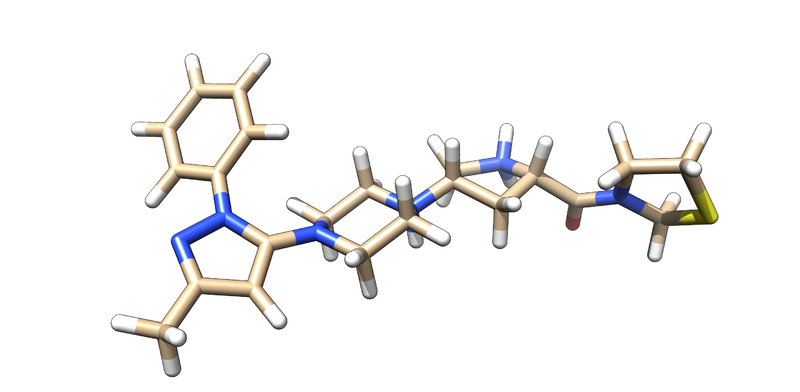
Preparing Ligand We will now need to prepare the Ligand, M-513. In a similar manner to receptor preparation, open the PDB file on Chimera. Likewise, you will also need to delete Chain B as previously stated. Now, you will be able to isolate the ligand by doing the following:
select->residue->M-51 select->invert Actions->Atoms/Bonds->Delete
You should be left with the following:
Next, we will save this as a mol2 file:
File->save as mol2 ->3VJK_ligand_noH.mol2
Add Hydrogens and Charge The crystal structure does not have any hydrogens because of technical limitations; hydrogen electron densities are too small to be detected. Consequently, we must add hydrogens to the ligand.
Tools->Structure editing-> add H
In a similar fashion, DOCK will need charges to perform calculations.
Tools->Structure editing-> Add Charge
It is important to make a note about the net charge of the ligand. you should not assume that chimera has the correct charge''. You should look at the ligand and attempt to validate the charge, which should be +2. You can now save this as a mol2 file and name it: 3VJK_ligand_with_H.mol2
Surface Generation & Sphere Selection
Surface Generation In Chimera a file which represents the surface of the protein will be created. The surface will be used to create a negative image of the protein (spheres which occupy the cavities and external face of the protein). These spheres are used to guide the ligand during docking.
In Chimera open "3VJK_rec_woH.mol2" :
Actions -> Surface -> Show Tools -> Structure Editing -> Write DMS -> "3VJK_rec_surface.dms"
Move this to the directory "002.surface_spheres"
Sphere Selection
By this step, you should have the mol2 extractions of ligand and protein, in both hydrogenated and unhydrogenated forms (4 files). The next activity is to create an efficient representation of empty space inside the protein. This is done with the sphgen script, which tries to generate the largest possible sphere for any given empty space. In general, it is desirable for the spheres will eclipse with each other, but not with the protein itself.
The sphgen software takes in a series of inputs from prompts to the user, but we can automate this by piping these arguments through a file. We shall can this file INSPH. Generate your INSPH file with the following syntax:
[your_receptor].dms <R flag> - enables sphere generation outside the protein surface (no eclipsing) <X flag - uses all coordinates <double> - distance that steric interactions are checked (units?) <double> - Maximum sphere radius of generated sphere (units?) <double> - Size of sphere that rolls over dms file surface for cavities (units?) [your_receptor].sph
This is an example of how we wrote our file:
3VJK_rec_surface.dms R X 0.0 4.0 1.4 3VJK_receptor_woH.sph
Does it matter if the dms is generated with the hydrogens?
This should produce an sph file that you can then run through sphgen
sphgen -i INSPH -o OUTSPH
Sphere Selection
Using dock's sphere_selector script, we are able to produce a subset of spheres that are close (within 10 angstroms) to the ligand
sphere_selector 3vjk_receptor.sph 3vjk_ligand_H.mol2 10.0
Generation of the Box and Grid
Energy calculations can be computationally expensive. Consequently, steps must be performed in order to reduce the number of calculations that are performed. In more detail, DOCK will be calculating the energy using a grid. We will be generating the grid; anything that is beyond the grid generated will not be in the calculation. This means that will ignore long distant interactions with ligand.
To start, we will be making a directory for the grid and the box
mkdir 003.gridbox
Generating the box
Next, we will be creating an input file that contains information for the Showbox programs. This file will contain parameters for the box.
vi showbox.in
we will put the following in to the file:
Y #generate box# 8.0 #how many angstroms the box edges should be from the spheres# ./../002.surface_spheres/select_spheres.sph #the location of the selected spheres# 1 3VJK.box.pdb #name of the output file#
In order to run this you do this:
showbox < showbox.in
After you run this command a file called 3VJK.box.pdb will be generated. This files contains the grid. And can be visualized.
Generating the grid
In a similar manner, we will have to generate the grid. In order to do this we will need to make the input file for the grid program that contains:
compute_grids yes grid_spacing 0.4 output_molecule no contact_score no energy_score yes energy_cutoff_distance 9999 atom_model a attractive_exponent 6 repulsive_exponent 9 distance_dielectric yes dielectric_factor 4. bump_filter yes bump_overlap 0.75 receptor_file d box_file d.box.pbd vdw_definition_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn score_grid_prefix grid
we will called this file grid.in. In order to generate the grid do the following:
grid -i grid.in -o gridinfo.out
the "-o" flag is used to specify the name of the output file. Once the program is completed there should be three files generated: gridinfo.out, grid.nrg, and grid.bmp. It is a good idea to make sure that gridinfo.out matches with the known information of the system. In other words, this is a good spot to double check your work.
Energy Minimization
Before running any dock calculations, we must take a moment to minimize the ligand. This is important because the current state of the ligand may not be at its lowest energy. We must take into consideration that crystallization can result in packing and other discrepancies that can impact our results. By minimizing the structure, we can make sure that none of the byproducts of crystalization will impact the results of the calculation.
We will make a new directory for Energy Minimization.
mkdir 004.energy_min
We will move into this directory. Now, we can conduct the first step to conducting energy minimization is to create an input file. We will call this file min.in:
conformer_search_type rigid use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ./../001.build/3VJK_ligand_hydrogens.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ./../001.build/3VJK_ligand_hydrogens.mol2 use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ./../003.gridbox/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes simplex_max_iterations 1000 simplex_tors_premin_iterations 0 simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_random_seed 0 simplex_restraint_min yes simplex_coefficient_restraint 10.0 atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl ligand_outfile_prefix 3VJK.lig.min write_orientations no num_scored_conformers 1 rank_ligands no
Now that our input file is made we can now start running minimization.
dock6 -i min.in -o min.out
once this command is run two files will be generated: min.out and 3VJK.lig.min.mol2. The mol2 file that is generated can be visualized on chimera.
Short Cut Using Bash Scripting
Running the previous steps can become tedious when one is working with a massive set of systems. Moreover, the use of a bash script can help to keep a record of what parameters were used for an experiment. A quick way to run these steps is with the following script:
#!/bin/sh
echo PDB name
read pdb
echo receptor file with hydrogen
read receptor
echo receptor file DMA
read receptor_DMS
echo ligand file
read ligand
EOF
echo generating spheres
cat > INSPH << EOF
$receptor_DMS
R
X
0.0
4.0
1.4
${pdb}_receptor.sph
EOF
echo selecting spheres
sphgen -i INSPH -o OUTSPH
sphere_selector pdb_receptor.sph $ligand 10.0
#generate grid
echo generating grid
cat > showbox.in << EOF
Y
8.0
${pdb}_receptor.sph
1
${pdb}.box.pdb
EOF
showbox<showbox.in
cat > grid.in << EOF
compute_grids yes
grid_spacing 0.4
output_molecule no
contact_score no
energy_score yes
energy_cutoff_distance 9999
atom_model a
attractive_exponent 6
repulsive_exponent 9
distance_dielectric yes
dielectric_factor 4
bump_filter yes
bump_overlap 0.75
receptor_file $receptor
box_file pdb.box.pdb
vdw_definition_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn
score_grid_prefix grid
EOF
grid -i grid.in -o gridinfo.out
cat > min.in << EOF
conformer_search_type rigid
use_internal_energy yes
internal_energy_rep_exp 12
enternal_energy_cutoff 100.0
ligand_atom_file $ligand
limit_max_ligands no
skip_molecule no
read_mol_solvation no
calculate_rmsd yes
use_rmsd_reference_mol yes
rmsd_reference_filename $ligand
use_database_filter no
orient_ligand no
bump_filter no
score_molecules yes
contact_score_primary no
contact_score_secondary no
grid_score_primary yes
grid_score_secondary no
grid_score_rep_rad_scale 1
grid_score_vdw_scale 1
grid_score_es_scale 1
grid_score_grid_prefix grid
multigrid_score_secondary no
dock3.5_score_secondary no
continuous_score_secondary no
footprint_similarity_score_secondary no
pharmacophore_score_secondary no
descriptor_score_secondary no
gbsa_zou_score_secondary no
gbsa_hawkins_score_secondary no
SASA_score_secondary no
amber_score_secondary no
minimize_ligand yes
simplex_max_iterations 1000
simplex_tors_premin_iterations 0
simplex_max_cycles 1
simplex_score_converge 0.1
simplex_cycle_converge 1.0
simplex_trans_step 1.0
simplex_rot_step 0.1
simplex_tors_step 10.0
simplex_random_seed 0
simplex_restraint_min yes
simplex_coefficient_restraint 10.0
atom_model all
vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn
flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn
flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl
ligand_outfile_prefix pdb.lig.min
write_orientations no
num_scored_conformers 1
rank_ligands no
EOF
echo minimized
dock6 -i min.in -o min.out
This can all be copied into a document named dock_setup.sh. This can become an executable file by using the command:
chmod u+x dock_setup.sh
Footprint Analysis
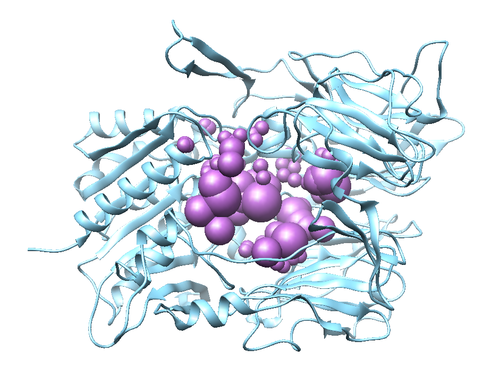
Using the electrostatic interactions and Van Der Waals Interactions, the molecular footprint can be used to understand how the ligand binds the receptor. Residues that are more negative are predominantly involved in the interaction.
First we will create a directory for the molecular footprint
mkdir 005.footprint
In this space, we will be creating the files that will be used for the molecular footprint. The first thing we have to do is make the footprint.in file:
conformer_search_type rigid use_internal_energy no ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd no use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary no grid_score_secondary no multigrid_score_primary no multigrid_score_secondary no dock3.5_score_primary no dock3.5_score_secondary no continuous_score_primary no continuous_score_secondary no footprint_similarity_score_primary yes footprint_similarity_score_secondary no fps_score_use_footprint_reference_mol2 yes fps_score_footprint_reference_mol2_filename ./../001.build/3VJK_ligand_hydrogens.mol2 fps_score_foot_compare_type Euclidean fps_score_normalize_foot no fps_score_foot_comp_all_residue yes fps_score_receptor_filename ./../001.build/3VJK_hydrogen_protein.mol2 fps_score_vdw_att_exp 6 fps_score_vdw_rep_exp 9 fps_score_vdw_rep_rad_scale 1 fps_score_use_distance_dependent_dielectric yes fps_score_dielectric 4.0 fps_score_vdw_fp_scale 1 fps_score_es_fp_scale 1 fps_score_hb_fp_scale 0 pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl ligand_outfile_prefix footprint.out write_footprints yes write_hbonds yes write_orientations no num_scored_conformers 1 rank_ligands no
Now you can use the following command to make the footprint:
dock6 -i footprint.in
Once this is completed you should have three new files: footprint.out_footprint_scored.txt footprint.out_hbond_scored.txt and footprint.out_scored.mol2
We can visualize the results using a python script: /gpfs/projects/AMS536/zzz.programs/plot_footprint_single_magnitude.py . This should be copied to the current directory.
cp /gpfs/projects/AMS536/2020/536_class/steve_ta/footprint_test_1.21.2020/plot_footprint_single_magnitude.py
This command can run by doing the following:
python plot_footprint_single_magnitude.py footprint.out_footprint_scored.txt 50
This will show the energetic distribution for the 50 most significant residues.
This distribution shows the relative stability of each residue with respect to the ligand before and after the docking process. In general, the docking process should arrange the ligand such that the global stability of the compound has increased.
Docking
Rigid Docking
This is the least computationally expensive docking method. This docking routine does not sample internal degrees of freedom (bond angles). The ligand is treated as a rigid object which is why minimization was performed in the prior step.
The first step is to create a dictory--where we will be putting are input and output files.
mkdir 006.rigid_docking
Now that we have the directory ready, we can make documents within this. The first thing we will want to do is set up the input file
touch rigid.in dock6 -i rigid.in
This will bring up an interactive input screen. Unlike the prior sets, it is highly suggested to manually complete this task because some options may be moved around depending on the version used. Below you can find the parameters that were used for this tutorial.
conformer_search_type flex write_fragment_libraries no user_specified_anchor no limit_max_anchors no min_anchor_size 5 pruning_use_clustering yes pruning_max_orients 1000 pruning_clustering_cutoff 100 pruning_conformer_score_cutoff 100.0 pruning_conformer_score_scaling_factor 1.0 use_clash_overlap no write_growth_tree no use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 use_database_filter no orient_ligand yes automated_matching yes receptor_site_file ./../002.surface_spheres/selected_spheres.sph max_orientations 1000 critical_points no chemical_matching no use_ligand_spheres no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ./../003.gridbox/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes minimize_anchor yes minimize_flexible_growth yes use_advanced_simplex_parameters no simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_anchor_max_iterations 500 simplex_grow_max_iterations 500 simplex_grow_tors_premin_iterations 0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl ligand_outfile_prefix rigid.out write_orientations no num_scored_conformers 1 rank_ligands no
Once the input is complete, there should be two output files: rigid.out and rigid.out_scored.mol2. Like the previous set with minimization, the results can be seen with chimera.
Fixed Anchor Docking
In this type of docking, the largest molecular fragment (the anchor) is oriented as the first step. The anchor is defined as the moiety containing no internal rotatable bonds possessing the most atoms. Subsequent rigid portions are attached and are oriented to be as energetically favorable as possible. This allows a larger space of sampling, but is still confined.
Make a directory called 007.fixed_anchor_docking/
In this directory, we will be creating an input file (like we did for the rigid docking) that is called flex.in
conformer_search_type flex write_fragment_libraries no user_specified_anchor no limit_max_anchors no min_anchor_size 5 pruning_use_clustering yes pruning_max_orients 1000 pruning_clustering_cutoff 100 pruning_conformer_score_cutoff 100.0 pruning_conformer_score_scaling_factor 1.0 use_clash_overlap no write_growth_tree no use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ./../003.gridbox/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes minimize_anchor yes minimize_flexible_growth yes use_advanced_simplex_parameters no simplex_max_cycles 1 simplex_score_converge .1 simplex_cycle_converge 1 simplex_trans_step 1 simplex_rot_step .1 simplex_tors_step 10.0 simplex_anchor_max_iterations 500 simplex_grow_max_iterations 500 simplex_grow_tors_premin_iterations 0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn
This can be run by doing the following:
dock6 -i fixed.in -o fixed.out
Once this is completed you should have two output files: flex.out and 2vjk_fad_scored.mol2.
Flexible Docking
Finally, this is the most expensive docking. This will sample all the internal degrees of freedom within the domain of bond angles.
Once again create a directory for this docking experiment. We will call it 008.flexible_docking
conformer_search_type flex write_fragment_libraries no user_specified_anchor no limit_max_anchors no min_anchor_size 5 pruning_use_clustering yes pruning_max_orients 1000 pruning_clustering_cutoff 100 pruning_conformer_score_cutoff 100.0 pruning_conformer_score_scaling_factor 1.0 use_clash_overlap no write_growth_tree no use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100.0 ligand_atom_file ./../004.energy_min/3VJK.lig.min_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd yes use_rmsd_reference_mol yes rmsd_reference_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 use_database_filter no orient_ligand yes automated_matching yes receptor_site_file ./../002.surface_spheres/selected_spheres.sph max_orientations 1000 critical_points no chemical_matching no use_ligand_spheres no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary yes grid_score_secondary no grid_score_rep_rad_scale 1 grid_score_vdw_scale 1 grid_score_es_scale 1 grid_score_grid_prefix ./../003.gridbox/grid multigrid_score_secondary no dock3.5_score_secondary no continuous_score_secondary no footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes minimize_anchor yes minimize_flexible_growth yes use_advanced_simplex_parameters no simplex_max_cycles 1 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_anchor_max_iterations 500 simplex_grow_max_iterations 500 simplex_grow_tors_premin_iterations 0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl ligand_outfile_prefix flex.out write_orientations no num_scored_conformers 1 rank_ligands no
Like the previous docking experiments we will do the following to make it run:
dock6 -i flex.in -o flex.out
Two files should have been generated: flex.out and flex.out_scored.mol2
Virtual Screening
Virtual screening is the process of passing a library of molecules through DOCK to a single receptor. This library of molecules can be tailored by the user to match the desired size and constituents. The advantage of a virtual screen is that many molecules can be analyzed with minimal user input, and the most strong binding molecules can be selected for further analysis. The screen performed for this tutorial used the library file "VS_library_5K.mol2" which can be found within the AMS536 directory. If desired to use a different library, all of the molecules must be provided within a single multi-mol2 file.
touch virtual.in
Input the following prompts:
conformer_search_type flex
write_fragment_libraries no
user_specified_anchor no
limit_max_anchors no
min_anchor_size 5
pruning_use_clustering yes
pruning_max_orients 1000
pruning_clustering_cutoff 100
pruning_conformer_score_cutoff 100.0
pruning_conformer_score_scaling_factor 1.0
use_clash_overlap no
write_growth_tree no
use_internal_energy yes
internal_energy_rep_exp 9
internal_energy_cutoff 100.0
ligand_atom_file VS_library_5K.mol2
limit_max_ligands no
skip_molecule no
read_mol_solvation no
calculate_rmsd no
use_database_filter no
orient_ligand yes
automated_matching yes
receptor_site_file ../002.surface_spheres/selected_spheres.sph
max_orientations 1000
critical_points no
chemical_matching no
use_ligand_spheres no
bump_filter no
score_molecules yes
contact_score_primary no
contact_score_secondary no
grid_score_primary yes
grid_score_secondary no
grid_score_rep_rad_scale 1
grid_score_vdw_scale 1
grid_score_es_scale 1
grid_score_grid_prefix ../003.gridbox/grid
multigrid_score_secondary no
dock3.5_score_secondary no
continuous_score_secondary no
footprint_similarity_score_secondary no
pharmacophore_score_secondary no
descriptor_score_secondary no
gbsa_zou_score_secondary no
gbsa_hawkins_score_secondary no
SASA_score_secondary no
amber_score_secondary no
minimize_ligand yes
minimize_anchor yes
minimize_flexible_growth yes
use_advanced_simplex_parameters no
simplex_max_cycles 1
simplex_score_converge 0.1
simplex_cycle_converge 1.0
simplex_trans_step 1.0
simplex_rot_step 0.1
simplex_tors_step 10.0
simplex_anchor_max_iterations 500
simplex_grow_max_iterations 500
simplex_grow_tors_premin_iterations 0
simplex_random_seed 0
simplex_restraint_min no
atom_model all
vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn
flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn
flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl
ligand_outfile_prefix virtual.out
write_orientations no
num_scored_conformers 1
rank_ligands no
Thus far all work has likely been done on a local machine. Running Dock on 5000 molecules could take an extended time if done on a local machine. To maximize the use of resources, a virtual screen can be done on the Seawolf Cluster. In order to do this, a submission script must be written which gives the cluster information on how to allocate resources.
Name the submission script virtual_screen.sh
vi virtual_screen.sh
#!/bin/sh
#SBATCH --partition=long-40core
#SBATCH --time=48:00:00
#SBATCH --nodes=1
#SBATCH --ntasks=1
#SBATCH --job-name=536.vs
#SBATCH --output=536.vs.out
dock6 -i virtual.in -o virtual.out
To submit the script in the bash shell:
sbatch virtual_screen.sh
You may check the status of you job:
squeue -u {username}
Once the job is completed you should read the output files.
cat 536.vs.out
Since only one node and 1 core, it is expected that the job was canceled due to reaching the time limit.
We can also see how many molecules were processed using this command:
grep Molecule: virtual_screening.out| wc -l
You may also visualize the results of the virtual screening by downloading virtual.out_scored.mol2 and using chimera
Tools-> Surface/BindingAnalysis->View Dock-> Dock6
This will allow you to visualize each molecule that was screened. Next, you can look at the properties of the molecules by downing the following:
Column->show
You can select from a variety of properties and rank the molecules based on these properties. This is useful for analyzing which molecules are favored in the active site
Consequently, we must re-run the job with a large number of nodes and cores using MPI.
Virtual Screening with MPI
In the previous section, it was found that the time limit was reached-- however--not all the molecules were screened. We simply cannot resubmit the job to complete the analysis. To compensate, we can submit the job to run on multiple cores. We can do this by using MPI.
You should first start off by making a new directory for the MPI Virtual Screening. This will be called 012.MPI_VS. You can simply copy the input file from the previous run. The only change will be in the submission script:
#!/bin/sh
#SBATCH --partition=long-40core
#SBATCH --time=48:00:00
#SBATCH --nodes=4
#SBATCH --ntasks=32
#SBATCH --job-name=tutorial_run
#SBATCH --output=%x-%j.o
echo "starting DOCK6.9 Simulation"
module load intel/mpi/64/2018/18.0.3
mpirun -np 32 dock6.mpi -i virtual_screening.in -o virtual_screening.out
echo "done"
In this script, I indicated that I will be using 4 nodes and a total of 32 cores. Additionally, it is required to load the module for mpi and use the dock6.mpi executable. Once the job has finish running you should find that a major of the molecules were calculated. Once again you may check using this command:
grep Molecule: virtual_screening.out| wc -l
In this tutorial, the 32 cores allowed for the calculation of 5665 out of the 5730 small molecules in the library. Thus, in the future, it may be required to allocate more resources to run through the whole library. Nevertheless, we may proceed using the available data.
Cartesian Minimization
We will now conduct minimization on the molecules that were docked. To do this, we must first create an input file:
conformer_search_type rigid use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100 ligand_atom_file ./../012.mpi_job/virtual.out_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd no use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary no grid_score_secondary no multigrid_score_primary no multigrid_score_secondary no dock3.5_score_primary no dock3.5_score_secondary no continuous_score_primary yes continuous_score_secondary no cont_score_rec_filename ./../001.build/3VJK_hydrogen_protein.mol2 cont_score_att_exp 6 cont_score_rep_exp 12 cont_score_rep_rad_scale 1 cont_score_use_dist_dep_dielectric yes cont_score_dielectric 4.0 cont_score_vdw_scale 1.0 cont_score_es_scale 1.0 footprint_similarity_score_secondary no pharmacophore_score_secondary no descriptor_score_secondary no gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand yes simplex_max_iterations 1000 simplex_tors_premin_iterations 0 simplex_max_cycles 1.0 simplex_score_converge 0.1 simplex_cycle_converge 1.0 simplex_trans_step 1.0 simplex_rot_step 0.1 simplex_tors_step 10.0 simplex_random_seed 0 simplex_restraint_min no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl ligand_outfile_prefix 3vjk.virtual_screen.min write_orientations no num_scored_conformers 1 rank_ligands no
We have named this min.in. It is important to make sure that the directory paths reflect your own paths. Once the input file has been made you should create a submission script:
#!/bin/sh #SBATCH --partition=long-40core #SBATCH --time=48:00:00 #SBATCH --nodes=3 #SBATCH --ntasks=24 #SBATCH --job-name=tutorial_run #SBATCH --dependency=afterok:227294 #SBATCH --output=%x-%j.o echo "starting Cartesian Minimization" dock6 -i min.in -o min.out echo "Minimization done"
You may name this job.sh. And now you should be able to submit the job.
In a similar manner to the previous section, you can download the output .mol2 files and load it using viewdock on chimera. This can be used to compare the unminimized ligands to the minimized ligands. You should expect to find that the energy is more negative after minimization.
Rescoring Molecules
We will now rescore the molecules based on several factors. This includes footprint, pharmacophore, Tanimoto, Hungarian, and volume_overlap score. The first step is to make a directory. This can be called rescore. Now you must make the input file (rescore.in):
conformer_search_type rigid use_internal_energy yes internal_energy_rep_exp 12 internal_energy_cutoff 100 ligand_atom_file ./../009.virtual_screening/virtual.out_scored.mol2 limit_max_ligands no skip_molecule no read_mol_solvation no calculate_rmsd no use_database_filter no orient_ligand no bump_filter no score_molecules yes contact_score_primary no contact_score_secondary no grid_score_primary no grid_score_secondary no multigrid_score_primary no multigrid_score_secondary no dock3.5_score_primary no dock3.5_score_secondary no continuous_score_primary no continuous_score_secondary no footprint_similarity_score_primary no footprint_similarity_score_secondary no pharmacophore_score_primary no pharmacophore_score_secondary no descriptor_score_primary yes descriptor_score_secondary no descriptor_use_grid_score no descriptor_use_multigrid_score no descriptor_use_continuous_score no descriptor_use_footprint_similarity yes descriptor_use_pharmacophore_score yes descriptor_use_hungarian yes descriptor_use_volume_overlap yes descriptor_fps_score_use_footprint_reference_mol2 yes descriptor_fps_score_footprint_reference_mol2_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 descriptor_fps_score_foot_compare_type Euclidean descriptor_fps_score_normalize_foot no descriptor_fps_score_foot_comp_all_residue yes descriptor_fps_score_receptor_filename ./../001.build/3VJK_hydrogen_protein.mol2 descriptor_fps_score_vdw_att_exp 6 descriptor_fps_score_vdw_rep_exp 12 descriptor_fps_score_vdw_rep_rad_scale 1 descriptor_fps_score_use_distance_dependent_dielectric yes descriptor_fps_score_dielectric 4.0 descriptor_fps_score_vdw_fp_scale 1 descriptor_fps_score_es_fp_scale 1 descriptor_fps_score_hb_fp_scale 0 descriptor_fms_score_use_ref_mol2 yes descriptor_fms_score_ref_mol2_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 descriptor_fms_score_write_reference_pharmacophore_mol2 no descriptor_fms_score_write_reference_pharmacophore_txt no descriptor_fms_score_write_candidate_pharmacophore no descriptor_fms_score_write_matched_pharmacophore no descriptor_fms_score_compare_type overlap descriptor_fms_score_full_match yes descriptor_fms_score_match_rate_weight 5.0 descriptor_fms_score_match_dist_cutoff 1.0 descriptor_fms_score_match_proj_cutoff .7071 descriptor_fms_score_max_score 20 descriptor_fingerprint_ref_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 descriptor_hms_score_ref_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 descriptor_hms_score_matching_coeff -5 descriptor_hms_score_rmsd_coeff 1 descriptor_volume_score_reference_mol2_filename ./../004.energy_min/3VJK.lig.min_scored.mol2 descriptor_volume_score_overlap_compute_method analytical descriptor_weight_fps_score 1 descriptor_weight_pharmacophore_score 1 descriptor_weight_fingerprint_tanimoto -1 descriptor_weight_hms_score 1 descriptor_weight_volume_overlap_score -1 gbsa_zou_score_secondary no gbsa_hawkins_score_secondary no SASA_score_secondary no amber_score_secondary no minimize_ligand no atom_model all vdw_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/vdw_AMBER_parm99.defn flex_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex.defn flex_drive_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/flex_drive.tbl chem_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/chem.defn pharmacophore_defn_file /gpfs/projects/AMS536/zzz.programs/dock6.9_release/parameters/ph4.defn ligand_outfile_prefix descriptor.output write_footprints yes write_hbonds yes write_orientations no num_scored_conformers 1 rank_ligands no
In a similar fashion to submitting the virtual screening, you may chose to submit the job using script:
#!/bin/sh #SBATCH --partition=long-40core #SBATCH --time=48:00:00 #SBATCH --nodes=4 #SBATCH --ntasks=32 #SBATCH --job-name=tutorial_run #SBATCH --output=%x-%j.o echo "starting DOCK6.9 Simulation" module load intel/mpi/64/2018/18.0.3 mpirun -np 32 dock6.mpi -i rescore.in -o rescore.out echo "done"
This can be submitted by doing the following
sbatch mpi.sh
Once the job has run you should be able to see the results by loading descriptor.output_scored.mol2 into chimera using view docks. You can select which scoring properties to look at by using the column feature.