Difference between revisions of "2019 DOCK tutorial 1 with PDBID 2BXF"
Stonybrook (talk | contribs) (→Selecting Spheres) |
Stonybrook (talk | contribs) |
||
| Line 106: | Line 106: | ||
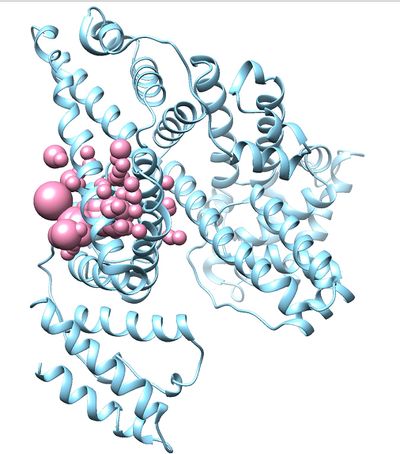
[[File:2BXF_receptor_and_selected_spheres.jpg|thumb|center|400px|2nnq receptor and selected spheres]] | [[File:2BXF_receptor_and_selected_spheres.jpg|thumb|center|400px|2nnq receptor and selected spheres]] | ||
| + | |||
| + | =IV. Generating box and grid= | ||
| + | |||
| + | ===Generating box=== | ||
| + | Move to 3.boxgrid directory | ||
| + | Create a new file showbox.in and write the following lines in the file. | ||
| + | |||
| + | Y | ||
| + | 8.0 | ||
| + | ../2.surface_spheres/selected_spheres.sph | ||
| + | 1 | ||
| + | 2nnq.box.pdb | ||
| + | |||
| + | Each of the above lines indicate that; | ||
| + | We intend to generate a box | ||
| + | The box length should be 8 Angstroms | ||
| + | Use the selected_spheres file in the designated location | ||
| + | The name of the file that contains generated box. | ||
| + | |||
| + | Use the following command to generate the box. | ||
| + | |||
| + | showbox < showbox.in | ||
| + | |||
| + | If this step is successful, you should see a new file (2nnq.box.pdb) in 3.boxgrid folder. | ||
| + | |||
| + | ===Generating grid=== | ||
| + | Create a new file (grid.in) | ||
| + | |||
| + | Use the following command to generate the grid. | ||
| + | grid -i grid.in -o gridinfo.out | ||
| + | |||
| + | Answer the prompted questions with the answers given below. '''(or you can use the following lines and include them in the grid.in file before entering the above command. If you do that these questions won't be prompted again. They will be automatically answered by grid.in file created)''' | ||
| + | |||
| + | compute_grids yes | ||
| + | grid_spacing 0.4 | ||
| + | output_molecule no | ||
| + | contact_score no | ||
| + | energy_score yes | ||
| + | energy_cutoff_distance 9999 | ||
| + | atom_model a | ||
| + | attractive_exponent 6 | ||
| + | repulsive_exponent 12 | ||
| + | distance_dielectric yes | ||
| + | dielectric_factor 4 | ||
| + | bump_filter yes | ||
| + | bump_overlap 0.75 | ||
| + | receptor_file ../1.dockprep/2nnq_rec_withH.mol2 | ||
| + | box_file 2nnq.box.pdb | ||
| + | vdw_definition_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/vdw_AMBER_parm99.defn | ||
| + | score_grid_prefix grid | ||
| + | |||
| + | If the command is successful, three new files will be generated. (gridinfo.out, grid.nrg, grid.bmp). Go through gridinfo.out file to make sure all the information about the receptor in the file matches with the original information of the receptor. (Eg:- Total charge, residues and their charges) If the information doesn't match, that means you have made an error in one of the steps that you followed so far. | ||
Revision as of 14:36, 18 February 2019
This tutorial contains a step by step approach to dock a known ligand to a known receptor.
Contents
I. Introduction
DOCK
DOCK is a molecular docking software that was originally developed at UCSF by Dr. Irwin Kuntz, Dr. Brian Shoicket, and colleagues. DOCK is used in drug discovery and molecular modeling. It consists of 2 main components: a search algorithm to explore chemical space for conformations and a scoring function to rank the results.
2BXF
The tutorial will be based on the PDB file 2BXF downloaded from the PDB Database. 2BXF is the crystal structure for human serum albumin complexed with diazepam.
Organization of Directories
We set up the files in our project space as such. It will be helpful to have these folders ready ahead of time.
0.files
1.dockprep
2.surface_spheres
3.gridbox
4.dock
6.footprint
7.virtual_screen
8.virtual_screen_mpi
9.cartesianmin
10.rescore
II. Preparation of the ligand and receptor
Download the pdb file 2BXF from Protein Data Bank and save the file in the 0.files folder.
Checking the Structure and Preparing the Complex without Hydrogens
- Open Chimera, open the downloaded pdb file, and check the structure for missing residues, gaps, heme groups, missing loops, and size. We recommend using proteins with no heme groups and proteins of a relatively smaller size. 2BXF did not have any of the preceding potential problems.
- Save the PDB file (the ligand and the receptor) as a mol2 file (2BXF_complex_noH.mol2) - When preparing both the ligand the receptor, you can open this complex mol2 file and delete the component that you won't need (eg. for preparing the receptor, open the complex, delete the ligand, and save).
Preparation of Receptor without Hydrogens
- Open 2BXF_complex_noH.mol2 through Chimera - Isolate the receptor by deleting the ligand. Click on the ligand and delete it (Actions -> Atoms/Bonds -> Delete). - Save the isolated receptor as a mol2 file (File -> Save mol2 -> 2BXF_rec_noH_mol2)
This receptor has been prepared without Hydrogens.
Preparation of Ligand without Hydrogens
- Open 2BXF_complex_noH.mol2 through Chimera again - Isolate the ligand by deleting the receptor. Click on the receptor and delete it (Actions -> Atoms/Bonds -> Delete). - Save the isolated ligand as a mol2 file (File -> Save mol2 -> 2BXF_lig_noH_mol2)
This ligand has been prepared without Hydrogens.
Preparation of receptor with Hydrogens
Preparation of ligand with Hydrogens
-Open the 2BXF_lig_noH.mol2 -Tools -> Surface Binding Analysis
Tools -> Structure Editing -> Add H (To add Hydrogen atoms)
Tools -> Structure Editing -> Add Charge (To add the charge use the latest AMBER force filed available for standard residues. Here we used AMBER ff14SB)
Save as a mol2 file. (2nnq_rec_withH.mol2)
- If you follow the step below all the above stated steps will automatically appear one after the other to prepare the receptor.
Tools -> Structure/Binding Analysis -> DockPrep
Preparation of ligand
- Open the PDB file via Chimera. - Using Chimera, isolate the ligand, add H atoms, add charge and save it as a mol2 file by following the same steps followed for the receptor.
Once all the files are prepared make sure to save the files in 1.dockprep folder.
III. Generating receptor surface and spheres
Preparation of DMS file
- Open 2BXF_rec_noH.mol2 using chimera. - Action -> Surface -> Show - Tools -> Structure Editing -> Write DMS - Save the 2BXF_rec_noH.dms into 3.surface_spheres folder
Reopen the file and make sure the surface was generated.
Transfer all the folders created so far to seawulf cluster to be used in DOCK.
Generating spheres
- Go to 2.surface_spheres folder - Create a new input file to create spheres by typing vim INSPH and type the following lines inside the file.
2BXF_rec_noH.dms R X 0.0 4.0 1.4 2BXF_rec.sph
The first line 2BXF_rec_noH.dms specifies the input file. R indicates that spheres generated will be outside of the receptor surface. X specifies all the points will be used. 0.0 is the distance in angstroms and it will avoid steric clashes. 4.0 is the maximum surface radius of the spheres and 1.4 is the minimum radius in angstroms.The last line 2BXF_spheres.sph creates the sph file that contains clustered spheres.
Once the INSPH file is ready, type the following command to generate the spheres.
sphgen -i INSPH -o OUTSPH
Once sphgen command is successful, 2BXF_spheres.sph file will be created. Open it up using Chimera along with 2BXF_rec_noH.mol2 file. You should get a similar output like the image below.
Selecting Spheres
Here we will be selecting the spheres which defines the binding pocket of the ligand because we are trying to direct the ligand towards that binding site rather than all over the receptor. To select the spheres type the following command.
sphere_selector 2BXF_rec.sph ../1.dockprep/2BXF_lig_withH.mol2 10.0
This command will select all of the spheres within 10.0 angstroms of the ligand and output them to selected_spheres.sph. Visualize the selected spheres using Chimera to make sure the correct spheres are selected. Notice that, spheres around the ligand binding site are kept and all the other spheres are deleted in the image below.
IV. Generating box and grid
Generating box
Move to 3.boxgrid directory Create a new file showbox.in and write the following lines in the file.
Y 8.0 ../2.surface_spheres/selected_spheres.sph 1 2nnq.box.pdb
Each of the above lines indicate that;
We intend to generate a box The box length should be 8 Angstroms Use the selected_spheres file in the designated location The name of the file that contains generated box.
Use the following command to generate the box.
showbox < showbox.in
If this step is successful, you should see a new file (2nnq.box.pdb) in 3.boxgrid folder.
Generating grid
Create a new file (grid.in)
Use the following command to generate the grid.
grid -i grid.in -o gridinfo.out
Answer the prompted questions with the answers given below. (or you can use the following lines and include them in the grid.in file before entering the above command. If you do that these questions won't be prompted again. They will be automatically answered by grid.in file created)
compute_grids yes grid_spacing 0.4 output_molecule no contact_score no energy_score yes energy_cutoff_distance 9999 atom_model a attractive_exponent 6 repulsive_exponent 12 distance_dielectric yes dielectric_factor 4 bump_filter yes bump_overlap 0.75 receptor_file ../1.dockprep/2nnq_rec_withH.mol2 box_file 2nnq.box.pdb vdw_definition_file /gpfs/projects/AMS536/zzz.programs/dock6/parameters/vdw_AMBER_parm99.defn score_grid_prefix grid
If the command is successful, three new files will be generated. (gridinfo.out, grid.nrg, grid.bmp). Go through gridinfo.out file to make sure all the information about the receptor in the file matches with the original information of the receptor. (Eg:- Total charge, residues and their charges) If the information doesn't match, that means you have made an error in one of the steps that you followed so far.