Difference between revisions of "2012 AMBER Tutorial with Biotin and Streptavidin"
Stonybrook (talk | contribs) (→MMGBSA Energy Calculation) |
Stonybrook (talk | contribs) (→Tuoling) |
||
| Line 508: | Line 508: | ||
===Tuoling=== | ===Tuoling=== | ||
| + | '''Some important things when modeling protein-protein interaction in AMBER''' | ||
| + | |||
| + | In structural preparation step, you should treat both proteins as receptors. Remove the water molecules and other small molecules needed. (Do not add hydrogens and charges in this case). Save the file into '''pdb''' fomat as Woo Suk wrote. You may need to modify the some pdb files. E.g. change residue name '''CYS''' to '''CYX''', change '''SH''' to '''SG''' otherwise it will not be recognized by tleap. | ||
===Yuchen=== | ===Yuchen=== | ||
Revision as of 16:20, 15 March 2012
For additional Rizzo Lab tutorials see AMBER Tutorials.
In this tutorial, we will learn how to run a molecular dynamics simulation of a protein-ligand complex. We will then post-process that simulation by calculating structural fluctuations (with RMSD) and free energies of binding (MM-GBSA).
Contents
I. Introduction
AMBER
Amber - Assisted Model Building with Energy Refinement - is a suite of about multiple programs for perform macromolecular simulations. Amber11, the current version of Amber, includes newly released functionality such as PMEMD, particle mesh Ewald MD and soft-core Thermodynamics Integration MD. For the tutorial, we are using an older version which is AMBER10.
The Amber 10 Manual is the primary resource to get started with Amber10. (Tip: Using Adobe Acrobat to view the file, you can simply search the document for keywords such as the name of a simulation parameter, which saves much time.) In addition, Amber Tools User's Manual serves as another reference while using Amber tools. You can also read the manual for Amber11 on Amber11 and AmberTools Users' Manuals
Here are some programs in Amber
- LEaP: an preparing program for constructing new or modified systems in Amber. It consists of the functions of prep, link, edit, and parm for earlier version of Amber.
- ANTECHAMBER: in additional to LEap, this main Antechamber suite program is for preparing input files other than standard nucleic acids and proteins.
- SANDER: according to the Amber 10 manual, it is 'a basic energy minimizer and molecular dynamics program' that can be used to minimize, equilibrate and sample molecular conformations. And this is the program we mainly use in this tutorial to generate trajectory files of the molecular system.
- PMEMD: version of SANDER that has improved parallel scaling property and optimized speed.
- PTRAJ: an analysis program for processing trajectory files. One can use ptraj to rotate, translate the structures, evaluate geometrical features and so on.
There is a mailing list you could sign-up for, as an additional resource.
Biotin and Streptavidin
For information of the Biotin and Streptavidin system, see 2012 DOCK tutorial with Streptavidin.
Organizing Directories
While performing MD simulations, it is convenient to adopt a standard directory structure / naming scheme, so that files are easy to find / identify. For this tutorial, we will use something similar to the following:
~username/AMS536/AMBER-Tutorial/001.CHIMERA.MOL.PREP/
002.ANTE.TLEAP/
003.SANDER/
004.PTRAJ/
005.MMGBSA/
II. Structural Preparation
Preparation in Chimera
In this AMBER tutorial, we will use the same system with previous DOCK part. Chimera can directly get the structure by its PDB ID 1DF8. To begin with, we need three files under directory 001.CHIMERA.MOL.PREP.
1DF8.rec.lig.pdb:
1. To lower computational cost and make the system clear, we remove chain B of the dimer. Select - chain - B, Action - Atoms/Bonds - delete
2. Remove the water molecules. Select - residue - HOH, Action - Atoms/Bonds - delete
Then we separate the receiver and its ligand.
1DF8.rec.noH.pdb:
Select the ligand and delete it. Select - Residue - BTN, Action - Atoms/Bonds - delete
1DF8.lig.chimera.mol2:
1. Select the protein and delete it. Select - Residue - BTN, Select - Invert, Action - Atoms/Bonds - delete
2. Use Dock Prep to add hydrogens and charges(AM1-BCC) to the ligand. Tools - Structure Editing - Dock Prep
antechamber
A antechamber input file requires all the atom names to be unique and it only uses the first 3 characters as the name. So if we use 1DF8.lig.chimera.mol2 as the input file, it will cause errors("H102" and "H103" will have the same name "H10").
14 O3 26.9770 10.6020 12.2050 O.2 1 BTN201 -0.6531
15 N2 28.6480 12.1210 11.8210 N.pl3 1 BTN201 -0.4789
16 C4 28.9670 13.2060 10.9010 C.3 1 BTN201 0.0816
17 H102 32.0358 17.4077 15.9597 H 1 BTN201 0.0272
18 H103 30.6885 18.0473 15.0102 H 1 BTN201 0.0219
19 H92 30.5603 15.6243 15.3130 H 1 BTN201 -0.0094
20 H93 32.1288 15.4384 14.4982 H 1 BTN201 0.0384
21 H82 29.6624 16.7125 13.2433 H 1 BTN201 0.0275
We need to manually rename the atoms. One way is to use the first column numbers to be the atom names. If you are using Vim, the visual block mode can help by selecting a rectangular section of text. We rename the atoms and save the file as 1DF8.lig.mol2 under directory 001.CHIMERA.MOL.PREP.
14 O14 26.9770 10.6020 12.2050 O.2 1 BTN201 -0.6531
15 N15 28.6480 12.1210 11.8210 N.pl3 1 BTN201 -0.4789
16 C16 28.9670 13.2060 10.9010 C.3 1 BTN201 0.0816
17 H17 32.0358 17.4077 15.9597 H 1 BTN201 0.0272
18 H18 30.6885 18.0473 15.0102 H 1 BTN201 0.0219
19 H19 30.5603 15.6243 15.3130 H 1 BTN201 -0.0094
20 H20 32.1288 15.4384 14.4982 H 1 BTN201 0.0384
21 H21 29.6624 16.7125 13.2433 H 1 BTN201 0.0275
LEaP
To begin with, go to 002.ANTE.TLEAP directory.
To make sure we have access to the three programs that we want to run (antechamber, parmchk and tleap) and we are using the correct version of amber, we can use the which command, type:
which antechamber which parmchk which tleap
You should have results similar to this:
/nfs/user03/tbalius/amber10_ompi/bin/antechamber /nfs/user03/tbalius/amber10_ompi/bin/parmchk /nfs/user03/tbalius/amber10_ompi/bin/tleap
Please note that you need to be in the tcsh shell for running the which command shown above. Also, if you are using a different version of amber, you might want to change to the correct version by editing the .cshrc file and source it.
Copy parameters of ions to your working directory from the following resource:
scp -r ~lingling/AMS536/AMBER_Tutorial/002.ANTE.TLEAP/rizzo_amber7.ionparms .
Antechamber is a set of tools to generate files for organic molecules, which can then be read into LEaP. The antechamber program itself is the main program of Antechamber package. It can perform many file conversions, and can also assign atomic charges and atom types. In this tutorial, we use antechamber to convert our input mol2 file into files ready for LEaP. In the command line, type:
antechamber -i ../001.CHIMERA.MOL.PREP/1DF8.lig.mol2 -fi mol2 -o 1DF8.lig.ante.pdb -fo pdb
-i input file name; -fi input file format; -o output file name; -fo output file format. You will have an output file:
1DF8.lig.ante.pdb
Similarly, we can use antechamber to change the fomat of 1DF8.lig.mol2 file to prep file:
antechamber -i ../001.CHIMERA.MOL.PREP/1DF8.lig.mol2 -fi mol2 -o 1DF8.lig.ante.prep -fo prepi
You will get a set of output files:
ANTECHAMBER_BOND_TYPE.AC ATOMTYPE.INF 1DF8.lig.ante.prep ANTECHAMBER_BOND_TYPE.AC0 NEWPDB.PDB ANTECHAMBER_AC.AC ANTECHAMBER_PREP.AC PREP.INF ANTECHAMBER_AC.AC0 ANTECHAMBER_PREP.AC0
These files give detailed information of the ligand such as atom coordinates, bond angle, bond length, dihedral angles, etc. For example:
vim ATOMTYPE.INF
You can see this file contains information related to the ring system.
Parmchk is another program included in the Antechamber package. After running antechamber, we run parmchk to check the parameters. If there are missing parameters after antechamber is finished, the frcmod template generated by parmchk will help us in generating the needed parameters:
parmchk -i 1DF8.lig.ante.prep -f prepi -o 1DF8.lig.ante.frcmod
Then open the output file 1DF8.lig.ante.frcmod, you will see something like this:
remark goes here MASS BOND ANGLE DIHE IMPROPER c3-o -c -o 1.1 180.0 2.0 General improper torsional angle (1 general atom type) n -n -c -o 10.5 180.0 2.0 General improper torsional angle (2 general atom types) NONBON
This means all the needed force field parameters are avalaible except there are two missing dihedral parameters, which were assigned a default value.
Next, we need create 3 new input files – “tleap.lig.in”, “tleap.rec.in” and “tleap.com.in”, for the ligand, the receptor, and the complex, respectively. These input files will be used by LEaP to create parameter/topology files and initial coordinate files.
vim tleap.lig.in
In this new ligand input file, type:
set default PBradii mbondi2 #set default PBradii source leaprc.ff99SB #load a force field loadoff rizzo_amber7.ionparms/ions.lib loadamberparams rizzo_amber7.ionparms/ions.frcmod #load an AMBER format parameter file source leaprc.gaff loadamberparams rizzo_amber7.ionparms/gaff.dat.rizzo loadamberparams 1DF8.lig.ante.frcmod loadamberprep 1DF8.lig.ante.prep #load an AMBER PREP file lig = loadpdb 1DF8.lig.ante.pdb #load the ligand pdb file saveamberparm lig 1DF8.lig.gas.leap.prm7 1DF8.lig.gas.leap.rst7 #save the ligand gas phase AMBER topology and coordinate files solvateBox lig TIP3PBOX 10.0 #solvate the receptor using TIP3P, solvent box radii 10 angstroms saveamberparm lig 1DF8.lig.wat.leap.prm7 1DF8.lig.wat.leap.rst7 #save the ligand water phase AMBER topology and coordinate files charge lig #charge the ligand quit
Similarly, create the tleap.rec.in and tleap.com.in files:
set default PBradii mbondi2 source leaprc.ff99SB loadoff rizzo_amber7.ionparms/ions.lib loadamberparams rizzo_amber7.ionparms/ions.frcmod REC = loadpdb ../001.CHIMERA.MOL.PREP/1DF8.rec.noh.pdb saveamberparm REC 1DF8.rec.gas.leap.prm7 1DF8.rec.gas.leap.rst7 solvateBox REC TIP3PBOX 10.0 saveamberparm REC 1DF8.rec.wat.leap.prm7 1DF8.rec.wat.leap.rst7 charge REC quit
set default PBradii mbondi2
source leaprc.ff99SB
loadoff rizzo_amber7.ionparms/ions.lib
loadamberparams rizzo_amber7.ionparms/ions.frcmod
source leaprc.gaff
loadamberparams rizzo_amber7.ionparms/gaff.dat.rizzo
REC = loadpdb ../001.CHIMERA.MOL.PREP/1DF8.rec.noh.pdb
loadamberparams 1DF8.lig.ante.frcmod
loadamberprep 1DF8.lig.ante.prep
LIG = loadpdb 1DF8.lig.ante.pdb
COM = combine {REC LIG}
saveamberparm COM 1DF8.com.gas.leap.prm7 1DF8.com.gas.leap.rst7
solvateBox COM TIP3PBOX 10.0
saveamberparm COM 1DF8.com.wat.leap.prm7 1DF8.com.wat.leap.rst7
charge COM
quit
Use tleap command:
tleap -s -f tleap.lig.in > tleap.lig.out tleap -s -f tleap.rec.in > tleap.rec.out tleap -s -f tleap.com.in > tleap.com.out
you will see the following output files:
1DF8.lig.gas.leap.prm7 1DF8.lig.gas.leap.rst7 1DF8.lig.wat.leap.prm7 1DF8.lig.wat.leap.rst7 1DF8.rec.gas.leap.prm7 1DF8.rec.gas.leap.rst7 1DF8.rec.wat.leap.prm7 1DF8.rec.wat.leap.rst7 1DF8.com.gas.leap.prm7 1DF8.com.gas.leap.rst7 1DF8.com.wat.leap.prm7 1DF8.com.wat.leap.rst7 tleap.lig.out tleap.rec.out tleap.com.out leap.log
An alternative way to use antechamber, parmchk and tleap is to make a csh script, which contains the commands you want to do, and run the script.
You can visualize the output files in VMD, and use the output files to run SANDER.
Reference:
2011 AMBER Tutorial with Biotin and Streptavidin
Visualization in VMD
First, copy “002.ANTE.TLEAP”directory back to Herbie. On Herbie,type the following commands:
cd ~/AMS536/AMBER_Tutorial/ scp –r sw:~/AMS536/AMBER_Tutorial/002.ANTE.TLEAP/ .
Open VMD software:
vmd
Then, we can load the gas phase ligand file generated by tleap: VMD Main->File->New Molecule (Please note that in order to visualize in VMD, we need first load the .prm7 file and then load the .rst7 file)
Load file for: New Molecule File name: 1DF8.lig.gas.leap.prm7 Determine file type: AMBER7 Parm
Load file for: 1DF8.lig.gas.leap.prm7 File name: 1DF8.lig.gas.leap.rst7 Determine file type: AMBER7 Restart
You can see the ligand in the gas phase looks like this:
You can compare this with ligand in the water phase by opening 1DF8.lig.water.leap.prm7, 1DF8.lig.water.leap.rst7 at the same time.
In order to see the ligands clearly, we can visualize without hydrogen atoms. VMD Main->Graphics->Representations->
In the Selected Atoms command line, type: all and noh->Click Apply
You can actually see the ligand in water is very different from that in gas phase.
III. Simulation using sander
Minimization and equilibration
Production
Although we run minimization and production in the same script, they represent distinct stages
We should run the production phase of the simulation using the same conditions as the final phase of equilibration to prevent an abrupt jump in the potential energy due to a change in simulation conditions.
First create the file 10md.in:
10md.in: production (500000 = 1ns)
&cntrl
imin = 0, ntx = 5, irest = 1, nstlim = 500000,
temp0 = 298.15, tempi = 298.15, ig = 71287,
ntc = 2, ntf = 1, ntt = 1, dt = 0.002,
ntb = 2, ntp = 1, tautp = 1.0, taup = 1.0,
ntwx = 500, ntwe = 0, ntwr = 500, ntpr = 500,
scee = 1.2, cut = 8.0, iwrap = 1,
ntr = 1, nscm = 100,
restraintmask = ':1-118@CA,C,N', restraint_wt = 0.1,
/
Then create similar files with the modified parameters:
06md.in: ntx = 1, irest = 0, nstlim = 50000, restraintmask = ':1-119 & !@H=',
restraint_wt = 1.0, dt = 0.001,
ntwx = 1000, ntwr = 1000, ntpr = 1000,
07md.in: nstlim = 50000, restraintmask = ':1-119 & !@H=', restraint_wt = 0.5, dt = 0.001,
ntwx = 1000, ntwr = 1000, ntpr = 1000,
08md.in: nstlim = 50000, restraintmask = ':1-118@CA,C,N', restraint_wt = 0.1, dt = 0.001,
ntwx = 1000, ntwr = 1000, ntpr = 1000,
09md.in: nstlim = 50000, restraintmask = ':1-118@CA,C,N', restraint_wt = 0.1, dt = 0.001,
ntwx = 1000, ntwr = 1000, ntpr = 1000,
10md.in: nstlim = 500000, restraintmask = ':1-118@CA,C,N', restraint_wt = 0.1, dt = 0.002, 11md.in: nstlim = 500000, restraintmask = ':1-118@CA,C,N', restraint_wt = 0.1, dt = 0.002,
Here the 10md.in and 11md.in use the pre-equilibrated system.
In The above code we see that we are running for 50000 steps (nstlim = 50000), with a time step of dt where dt of 0.001 is 1fs. We are starting the run with no previous velocity information ntx=1, irest=0. We will print energy output every 500 steps (ntwr=500) and save coordinates every 500 (ntwx=500). We want the initial temperature to be 298.15K (tempi=298.15) and the reference temperature to be 300K (temp0=298.15). Initially, we restraint everything except hydrogens with a restraint weight of 1.0, and with later runs we decrease the restraint weight and only the backbone is restrained. With each future run, we are using velocity information from the previous restart file (ntx=5, irest=1), and switched on constant pressure (ntb=2) with isotropic position scaling (ntp=1).
We should keep in mind that each simulation stage is distinct, so if a simulation terminates prematurely, we can easily continue from the last completed stage using the -c flag. (e.g.,-c 10md.rst7).
To run equilibration and production, Write the tcsh file equil.produc.qsub.csh:
#! /bin/tcsh
#PBS -l nodes=4:ppn=2
#PBS -l walltime=720:00:00
#PBS -o zzz.qsub.out
#PBS -e zzz.qsub.err
#PBS -V
set workdir = "~/AMBER_Tutorial/003.SANDER"
cd ${workdir}
cat $PBS_NODEFILE | sort | uniq
mpirun -n 8 sander.MPI -O -i 01mi.in -o 01mi.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c ../002.TLEAP/1df8.com.wat.leap.rst7 -ref ../002.TLEAP/1df8.com.wat.leap.rst7 \
-x 01mi.trj -inf 01mi.info -r 01mi.rst7
mpirun -n 8 sander.MPI -O -i 02md.in -o 02md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 01mi.rst7 -ref 01mi.rst7 -x 02md.trj -inf 02md.info -r 02md.rst7
mpirun -n 8 sander.MPI -O -i 03mi.in -o 03mi.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 02md.rst7 -ref 02md.rst7 -x 03mi.trj -inf 03mi.info -r 03mi.rst7
mpirun -n 8 sander.MPI -O -i 04mi.in -o 04mi.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 03mi.rst7 -ref 03mi.rst7 -x 04mi.trj -inf 04mi.info -r 04mi.rst7
mpirun -n 8 sander.MPI -O -i 05mi.in -o 05mi.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 04mi.rst7 -ref 04mi.rst7 -x 05mi.trj -inf 05mi.info -r 05mi.rst7
mpirun -n 8 sander.MPI -O -i 06md.in -o 06md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 05mi.rst7 -ref 05mi.rst7 -x 06md.trj -inf 06md.info -r 06md.rst7
mpirun -n 8 sander.MPI -O -i 07md.in -o 07md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 06md.rst7 -ref 05mi.rst7 -x 07md.trj -inf 07md.info -r 07md.rst7
mpirun -n 8 sander.MPI -O -i 08md.in -o 08md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 07md.rst7 -ref 05mi.rst7 -x 08md.trj -inf 08md.info -r 08md.rst7
mpirun -n 8 sander.MPI -O -i 09md.in -o 09md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 08md.rst7 -ref 05mi.rst7 -x 09md.trj -inf 09md.info -r 09md.rst7
mpirun -n 8 sander.MPI -O -i 10md.in -o 10md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 09md.rst7 -ref 05mi.rst7 -x 10md.trj -inf 10md.info -r 10md.rst7
mpirun -n 8 sander.MPI -O -i 11md.in -o 11md.out -p ../002.TLEAP/1df8.com.wat.leap.prm7 \
-c 10md.rst7 -ref 05mi.rst7 -x 11md.trj -inf 11md.info -r 11md.rst7
exit
Submit the file to the que and monitor progress.
IV. Simulation Analysis
Ptraj
Yuchen
To run the Ptraj analysis, you will probably first create a separate folder for it under the root directory if you don't have one.
mkdir 004.PTRAJ
We will run five Ptraj analysis so that we need five separate input files. We will first create ptraj.1.in containing the following lines.
trajin ../003.SANDER/10md.trj 1 1000 1 trajin ../003.SANDER/11md.trj 1 1000 1 trajout 1DF8.trj.strip nobox strip :WAT
This will concatenate your two 1ns trajectories together, strip off the waters, and output it as a new file called 1DF8.trj.strip. The two sets of numbers 1 1000 1 give you the input information about which frames you are using for the Ptraj. The first two numbers 1 and 1000 specify the starting and ending snapshots from your trajectory file. The ending number of the snapshot doesn't need to be accurate because if you actually don't have enough snapshots in your trajectory file, Ptraj will read up to the last one you have. The last number 1 specifies the frequency of the snapshot saved, in this case, we are saving every frame of the trajectory file. And the last line of the input file will take away all the water molecules.
To execute the first Ptraj analysis, run the following command specifying both input and out put files.
ptraj ../002.ANTE.TLEAP/1DF8.com.wat.leap.parm ptraj.1.in > ptraj.1.log
After the first step, we now have the 1DF8.trj.strip file which contains 2000 snapshots of the trajectory, and we will compare it to our reference file 1DF8.com.gas.leap.rst7, using the following input file named as ptraj.2.in.
trajin 1DF8.trj.strip 1 2000 1 trajout 1DF8.com.trj.stripfit reference ../002.ANTE.TLEAP/1DF8.com.gas.leap.rst7 rms reference out 1DF8.rmsd.CA.txt :1-118@CA
Note that in this input file, the set of numbers for trajin is 1 2000 1 because in 1DF8.trj.strip we actually have 2000 snapshots. The third line specifies the reference file we are using as 1DF8.com.gas.leap.rst7, because we have taken away all the water molecules in the first step, we are comparing our trajectory to the gas phase complex. And the last line says we are calculating the rmsd for alpha carbon number 1 to 118.
To execute the the analysis, run the following line.
ptraj ../002.ANTE.TLEAP/1DF8.com.gas.leap.parm ptraj.2.in > ptraj.2.log
This will give us an txt file with two columns in it, the first column is the number of frame it is comparing and the second column is the rmsd. And then we will generate a similar rmsd file for ligand as well using the input file as follows.
trajin 1DF8.com.trj.stripfit 1 2000 1 reference ../002.ANTE.TLEAP/1DF8.com.gas.leap.rst7 rms reference out 1DF8.lig.rmsd.txt :119@C*,N*,O*,S* nofit
Similarly, this time we will compare the trajectory file to 1DF8.com.gas.leap.rst7 as well, only this time we are comparing the ligand instead of the receptor by specifying that in the last line saying we want to calculate the rmsd for carbon, nitrogen, oxygen and sulfur in residue 119. And also the <nofit> in the last line specifying that we are calculating the rmsd of the ligand to the crystal structure without any fitting.
To execute the the analysis, run the following line.
ptraj ../002.ANTE.TLEAP/1DF8.com.gas.leap.parm ptraj.3.in > ptraj.3.log
Woo Suk
Finally, you want to perform MMGBSA analysis to obtain energetic information about the system. To do this, you need a trajectory file of the gas phase complex (which we already created above and saved as 1DF8.com.trj.stripfit), and want to create two more trajectory files containing just the receptor and just the ligand. To do so, you have to run two more ptraj using the following ptraj input files:
ptraj.4.in
trajin 1DF8.com.trj.stripfit 1 2000 1 trajout 1DF8.rec.trj.stripfit strip :119
ptraj.5.in
trajin 1DF8.com.trj.stripfit 1 2000 1 trajout 1DF8.lig.trj.stripfit strip :1-118
To execute, type in the following commands:
ptraj ../002.ANTE.TLEAP/1DF8.com.gas.leap.parm ptraj.4.in > ptraj.4.log ptraj ../002.ANTE.TLEAP/1DF8.com.gas.leap.parm ptraj.5.in > ptraj.5.log
To visualize trajectories, we have to move the trajectory files to Herbie with scp or rsync commands and run VMD.
rsync -arv 004.PTRAJ/ ID@compute.mathlab.sunysb.edu:~AMS536/AMBER_tutorial/004.PTRAJ
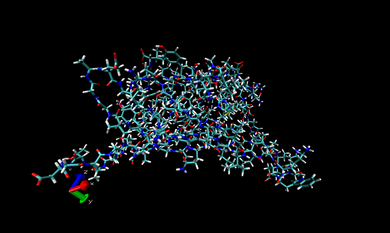
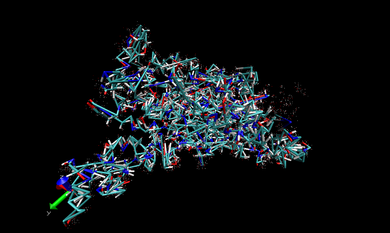
In VMD, we open one of the .prm7 files in 002.ANTE.TLEAP. If you want to visualize the whole complex in gas state, you can open 1DF8.com.gas.leap.prm7 with AMBER7 Parm from 002.ANTE.TLEAP and then 1DF8.com.trj.stripfit with AMBER coordinates from 004.PTRAJ. With these files, you can look at the real-time movement of the complex in the gas state like following snapshot.
You can repeat this procedure to observe the real-time movement of the complex in the water state. Only different thing is opening 1DF8.com.wat.leap.prm7 instead of 1DF8.com.gas.leap.prm7. The following snapshot is that of movement in the water phase.
In the snapshot above, the water molecules are shown as points.
MMGBSA Energy Calculation
MM/GBSA is the acronym for Molecular Mechanics/Poisson-Boltzmann Surface Area. This part of AMBER combines molecular mechanics (MM) with both the electrostatic (PB) and nonpolar (SA) contribution to solvation . Topology files are needed for the receptor (R), ligand (L), and receptor-ligand complex (RL). The trajectory files (trj) generate the coordinates. Therefore, molecular dynamics is used to generate a set of snapshots taken at fixed intervals from the trajectories. These snapshots are processed to remove solvent and generate the free energy of binding.
In the AMBER_Tutorial directory, create a new directory:
mkdir 005.MMGBSA
In this new directory, create the file gb.score.in containing:
Single point GB energy calc &cntrl ntf = 1, ntb = 0, ntc = 2, idecomp= 0, igb = 5, saltcon= 0.00, gbsa = 2, surften= 1.0, offset = 0.09, extdiel= 78.5, cut = 99999.0, nsnb = 99999, scnb = 2.0, scee = 1.2, imin = 5, maxcyc = 1, ncyc = 0, /
Then create a csh script, run.sander.rescore.csh, that contains the following lines of command:
#! /bin/tcsh
#PBS -l nodes=1:ppn=2
#PBS -l walltime=24:00:00
#PBS -o zzz.qsub.out
#PBS -e zzz.qsub.err
#PBS -V
set workdir = “${HOME}/AMBER_Tutorial/005.MMGBSA”
cd ${workdir}
sander -O -i gb.rescore.in \
-o gb.rescore.out.com \
-p ../002.TLEAP/1df8.com.gas.leap.prm7 \
-c ../002.TLEAP/1df8.com.gas.leap.rst7 \
-y ../004.PTRAJ/1df8.com.trj.stripfit \
-r restrt.com \
-ref ../002.TLEAP/1df8.com.gas.leap.rst7 \
-x mdcrd.com \
-inf mdinfo.com
sander -O -i gb.rescore.in \
-o gb.rescore.out.lig \
-p ../002.TLEAP/1df8.lig.gas.leap.prm7 \
-c ../002.TLEAP/1df8.lig.gas.leap.rst7 \
-y ../004.PTRAJ/1df8.lig.trj.stripfit \
-r restrt.lig \
-ref ../002.TLEAP/1df8.lig.gas.leap.rst7 \
-x mdcrd.lig \
-inf mdinfo.lig
sander -O -i gb.rescore.in \
-o gb.rescore.out.rec \
-p ../002.TLEAP/1df8.rec.gas.leap.prm7 \
-c ../002.TLEAP/1df8.rec.gas.leap.rst7 \
-y ../004.PTRAJ/1df8.rec.trj.stripfit \
-r restrt.rec \
-ref ../002.TLEAP/1df8.rec.gas.leap.rst7 \
-x mdcrd.rec \
-inf mdinfo.rec
exit
Then this script should be sent to the queue, i.e., qsub the script using the commands:
chmod +x run.sander.rescore.csh qsub run.sander.rescore.csh
You can monitor your progress by typing qstat -u username.
When the job is complete, you will obtain the following output files: gb.rescore.out.com, gb.rescore.out.lig, and gb.rescore.out.rec
In these files, the single point energy calculation results will be written for each individual frame. It will be found in the results section and the output file will have an infrastrucutre that is similar to the following:
FINAL RESULTS
NSTEP ENERGY RMS GMAX NAME NUMBER
1 7.9662E+03 1.9504E+01 1.3272E+02 C 3403
BOND = 836.7674 ANGLE = 2343.1208 DIHED = 2917.9356
VDWAALS = -2299.0570 EEL = -19265.6215 EGB = -3539.4050
1-4 VDW = 1071.1234 1-4 EEL = 11631.2887 RESTRAINT = 0.0000
ESURF = 14270.0746
minimization completed, ENE= 0.79662270E+04 RMS= 0.195036E+02
minimizing coord set # 2
The results generated in the output files will be used for the final calculations. Use the grep command for complex (com), ligand (lig), and receptor (rec) to obtain the results.
In the command line, type:
grep VDWAALS gb.rescore.out.com > vdw.com.txt.
grep ESURF gb.rescore.out.com > surf.com.txt.
V. Frequently Encountered Problems
Barbara
1.If you have a problem opening the structure of your molecule using VMD, be certain that the prm7 file corresponds to the rst7 file. For example, 1df8.lig.wat.leap.prm7 must be matched to 1df8.lig.wat.leap.rst7.
2. The VMD Console window (often hidden behind the visualization screen of your molecule) can be used to determine where VMD has found an error in your typed commands if you are unable to visualize your molecule.
3. If your "rescore" files do not appear when you are running the script: run.sander.rescore.csh, it may be the result of there not being a free node. You job may be sitting on the queue. If you type qstat -u username, you will be able to see if the job is sitting there because a Q will appear.
Woo Suk
How to run DNA molecule in AMBER
If you have a DNA molecule as a substrate, you cannot run antechamber to add hydrogen atoms and charge. The antechamber is only for small molecules. In this case, you don't have to save ligand file as .mol2. Save the ligand file with pdb format and just run tleap like protein. Only different thing compared to protein tleap.in file is the force field. For DNA molecules, you have to use ff99bsc0 forcefield instead of ff99sb like following:
set default PBradii mbondi2 source leaprc.ff99bsc0 loadoff rizzo_amber7.ionparms/ions.lib loadamberparams rizzo_amber7.ionparms/ions.frcmod lig = loadpdb ../001.CHIMERA.MOL.PREP/3tmm.lig.pdb saveamberparm lig 3tmm.lig.gas.leap.prm7 3tmm.lig.gas.leap.rst7 solvateBox lig TIP3PBOX 10.0 saveamberparm lig 3tmm.lig.wat.leap.prm7 3tmm.lig.wat.leap.rst7 charge lig quit
In DNA pdb file, you have to note that the atom names must be DA, DT, DG and DC format. Your parameter file cannot recognize other name format like AD or dA.
Longfei
Roman
Quan
Hui
Tuoling
Some important things when modeling protein-protein interaction in AMBER
In structural preparation step, you should treat both proteins as receptors. Remove the water molecules and other small molecules needed. (Do not add hydrogens and charges in this case). Save the file into pdb fomat as Woo Suk wrote. You may need to modify the some pdb files. E.g. change residue name CYS to CYX, change SH to SG otherwise it will not be recognized by tleap.